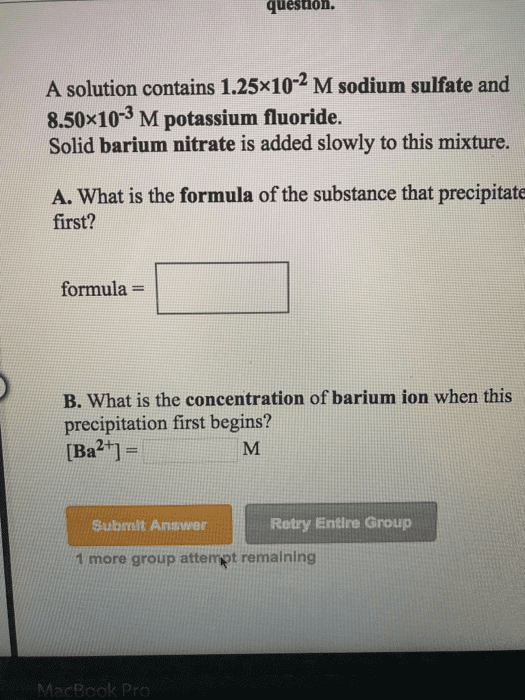
1
answer
0
watching
468
views
12 Dec 2019
A chemist attempts to seperate barium ions from lead ions by using the sulfate ion as a precipitating agent.
a) What sulfate ion concentrations are required for the precipitation of BaSO4 and pbSO4 from a solution containing 0.010M Ba^2+ aq and 0.010M Pb^2+ aq?
b) What is the concentration of barium ions when the lead sulfate begins to precipitate?
A chemist attempts to seperate barium ions from lead ions by using the sulfate ion as a precipitating agent.
a) What sulfate ion concentrations are required for the precipitation of BaSO4 and pbSO4 from a solution containing 0.010M Ba^2+ aq and 0.010M Pb^2+ aq?
b) What is the concentration of barium ions when the lead sulfate begins to precipitate?
Liked by cfmiara03
Bunny GreenfelderLv2
13 Dec 2019