1
answer
0
watching
291
views
12 Dec 2019
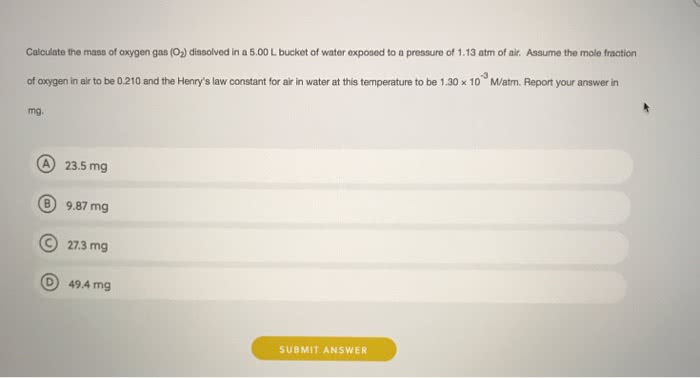
At 298 K, the Henry\'s law constant for oxygen is 0.00130 M/atm. Air is 21.0% oxygen.
At 298 K, what is the solubility of oxygen in water exposed to air at 1.00 atm? _____M
At 298 K, what is the solubility of oxygen in water exposed to air at 0.893 atm? ______M
If atmospheric pressure suddenly changes from 1.00 atm to 0.893 atm at 298 K, how much oxygen will be released from 5.00 L of water in an unsealed container? _______mol
At 298 K, the Henry\'s law constant for oxygen is 0.00130 M/atm. Air is 21.0% oxygen.
At 298 K, what is the solubility of oxygen in water exposed to air at 1.00 atm? _____M
At 298 K, what is the solubility of oxygen in water exposed to air at 0.893 atm? ______M
If atmospheric pressure suddenly changes from 1.00 atm to 0.893 atm at 298 K, how much oxygen will be released from 5.00 L of water in an unsealed container? _______mol
Collen VonLv2
13 Dec 2019