1
answer
0
watching
742
views
12 Dec 2019
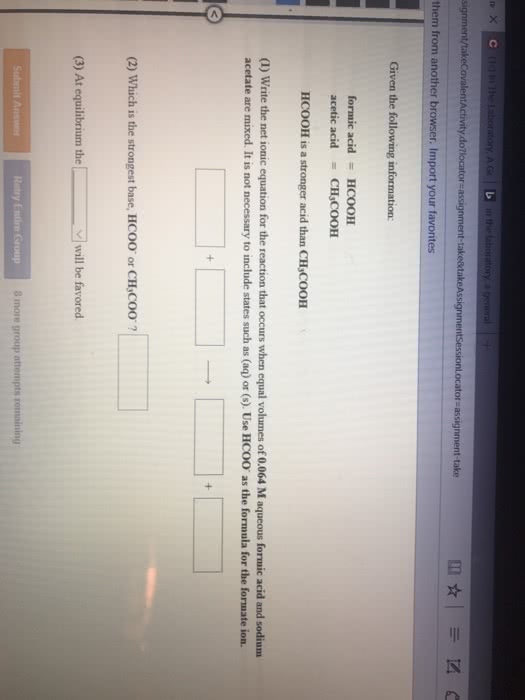
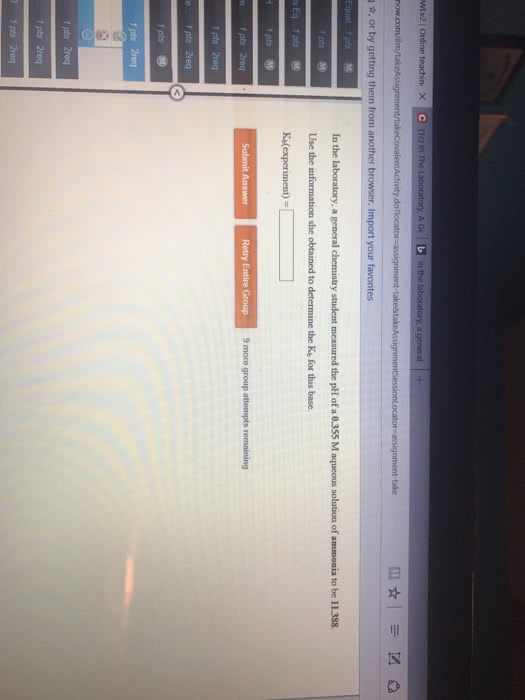
Given the following information:
hypochlorous acid = HClO formic acid = HCOOH
HCOOH is a stronger acid than HClO
(1) Write the net ionic equation for the reaction that occurs when equal volumes of 0.090 M aqueous hypochlorous acid and sodiumformate are mixed. It is not necessary to include states such as (aq) or (s). Use HCOO- as the formula for the formate ion.
(2) Which is the strongest base, ClO-or HCOO-?
3) At equilibrium the _____ will be favored. (reactants or products)
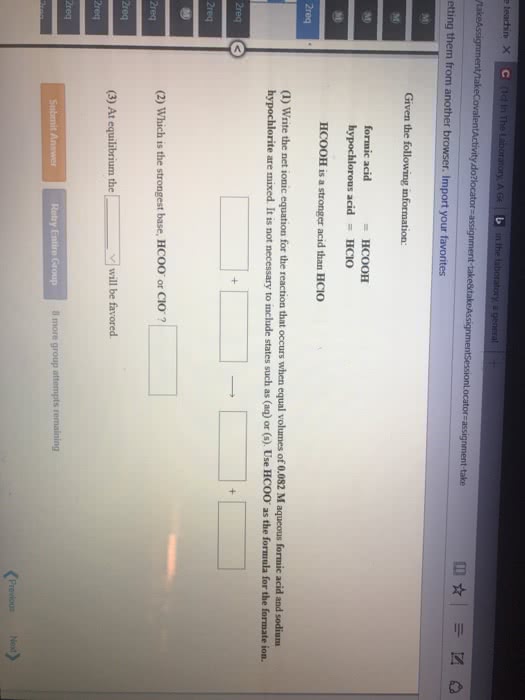
Given the following information:
| hypochlorous acid | = | HClO |
| formic acid | = | HCOOH |
HCOOH is a stronger acid than HClO
(1) Write the net ionic equation for the reaction that occurs when equal volumes of 0.090 M aqueous hypochlorous acid and sodiumformate are mixed. It is not necessary to include states such as (aq) or (s). Use HCOO- as the formula for the formate ion.
| (2) Which is the strongest base, ClO-or HCOO-? 3) At equilibrium the _____ will be favored. (reactants or products) |
Nelly StrackeLv2
13 Dec 2019