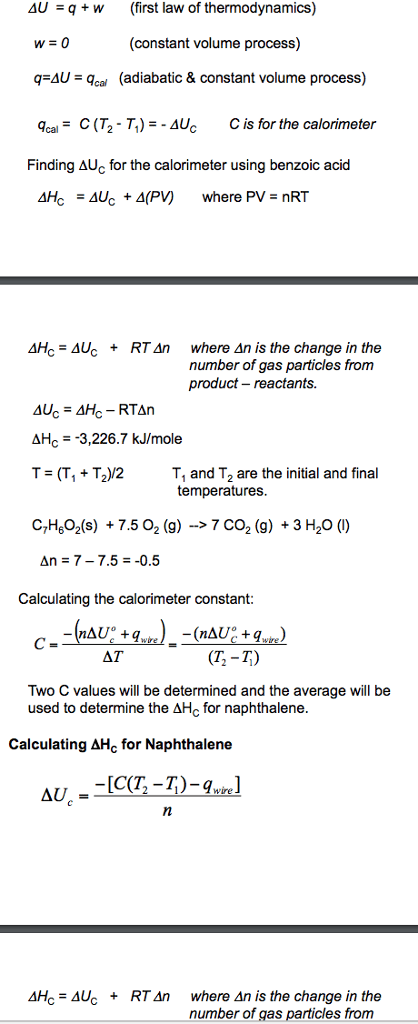1
answer
0
watching
214
views
12 Dec 2019
so I have following data but I don't know how to calculateforq temperature of HCL 22.2 C temperature of NaOH 21.9 C Final temperature of resulting salt solution (Tf) 22.6 C q=mcÎT so m = 100 g c=4.18 j g/C ÎT=30.4 - ? what is delta T? and after that how do you calculate molesofNaOH or HCL netrualized (moles of rxn) also how do u calculate: Enthalpy change (using expt data) for OH- + H+ --->H2O(l) standard enthalpy of formation H2O Calculated enthalpy of formation of OH- please help me!! ------ Hello guys. I have to do these calculations but I can seemtodo it and I am stuck. So can someone please help me calculatethesevalues? We know that the specific heat of water (same for solutionforthis expt) is 4.18 J/g C and densities of all solutions are 1.00 g/mL Temp of HCl: 22.2 C Temp of NaOH: 22.0 C so initial Temp is average of those two which is 22.1 C final temperature of the resulting salt solution: 22.6 so; q=mcÎT m= 100g c=4.18 J/g C ÎT= 22.6 - 22.1 = 0.5 C so q = (100)(4.18)(0.5) q= 209 J so q rxn = -q soln so =209 J ok now how do you calculate moles of NaOH or HCLneutralized (molesof rxn)once we get moles of rxn we can calculate theenthalpyof neutralization of rxn which is calculated by qrxn/(molesofrxn) so how do you calculate: enthalpy change (using expt data) for OH-+H+ -> H2O (l) standard enthalpy for formation H2O(l)(ÎH) calculated enthalpy for formation OH-(aq)(ÎH)
so I have following data but I don't know how to calculateforq
temperature of HCL 22.2 C
temperature of NaOH 21.9 C
Final temperature of resulting salt solution (Tf) 22.6 C
q=mcÎT
so m = 100 g
c=4.18 j g/C
ÎT=30.4 - ?
what is delta T? and after that how do you calculate molesofNaOH or HCL netrualized (moles of rxn)
also how do u calculate:
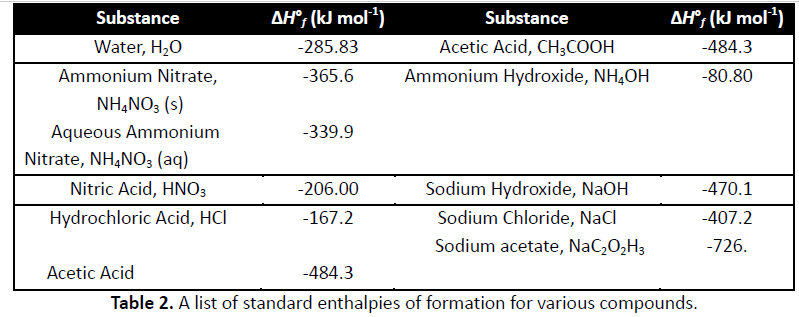
Enthalpy change (using expt data) for OH- + H+ --->H2O(l)
standard enthalpy of formation H2O
Calculated enthalpy of formation of OH-
please help me!!
------
Hello guys. I have to do these calculations but I can seemtodo it and I am stuck. So can someone please help me calculatethesevalues?
We know that the specific heat of water (same for solutionforthis expt) is 4.18 J/g C
and densities of all solutions are 1.00 g/mL
Temp of HCl: 22.2 C
Temp of NaOH: 22.0 C
so initial Temp is average of those two which is 22.1 C
final temperature of the resulting salt solution: 22.6
so;
q=mcÎT
m= 100g
c=4.18 J/g C
ÎT= 22.6 - 22.1 = 0.5 C
so q = (100)(4.18)(0.5)
q= 209 J
so q rxn = -q soln
so =209 J
ok now how do you calculate moles of NaOH or HCLneutralized (molesof rxn)
once we get moles of rxn we can calculate theenthalpyof neutralization of rxn
which is calculated by qrxn/(molesofrxn)
so how do you calculate:
enthalpy change (using expt data) for OH-+H+ -> H2O (l)
standard enthalpy for formation H2O(l)(ÎH)
calculated enthalpy for formation OH-(aq)(ÎH)
Jarrod RobelLv2
13 Dec 2019