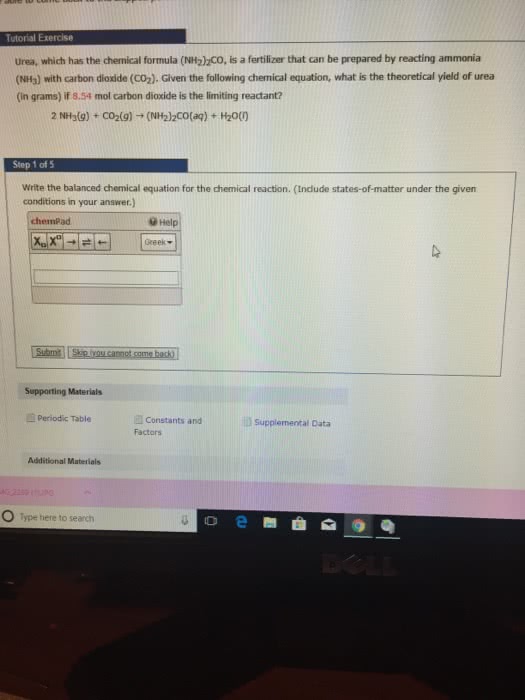
1
answer
0
watching
77
views
12 Dec 2019
Outline the steps needed to determine the limiting reactant when 27.4 g of propane, C3H8, is burned with 77.9 g of oxygen. Step 1: Write the balanced reaction equation. (Omit states-of-matter from your answer. Use the lowest possible whole number coefficients Step 2: Calculate the molar masses.Step 3: Determine the limiting reactant.Step 4: Calculate the theoretical yield of CO2 in grams
Outline the steps needed to determine the limiting reactant when 27.4 g of propane, C3H8, is burned with 77.9 g of oxygen. Step 1: Write the balanced reaction equation. (Omit states-of-matter from your answer. Use the lowest possible whole number coefficients Step 2: Calculate the molar masses.Step 3: Determine the limiting reactant.Step 4: Calculate the theoretical yield of CO2 in grams
Patrina SchowalterLv2
13 Dec 2019