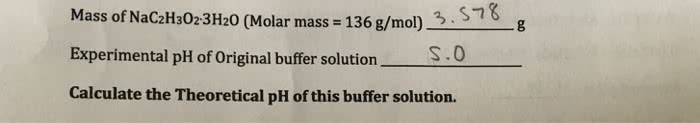1
answer
0
watching
235
views
12 Dec 2019
An acetic acid/ sodium acetate buffer solution was prepared using the following components:
3.46 g of NaC2H3O2â3H2O (FW. 136 g/mol)
9.0 mL of 3.0 M HC2H3O2
55.0 mL of water
-What is the total volume of the solution?
-Calculate the concentration of the [C2H3O2-] in this solution.
-Calculate the concentration of the [HC2H3O2] in this solution.
-Calculate the pH of this buffer solution. The Ka for acetic acid is 1.8x10-5.
-If you take half of this solution and add 2 mL of 1.00 M HCl to it, then what is the pH of this new solution?
-If you take half of this solution and add 2 mL of 1.00 M NaOH to it, then what is the pH of this new solution?
An acetic acid/ sodium acetate buffer solution was prepared using the following components:
3.46 g of NaC2H3O2â3H2O (FW. 136 g/mol)
9.0 mL of 3.0 M HC2H3O2
55.0 mL of water
-What is the total volume of the solution?
-Calculate the concentration of the [C2H3O2-] in this solution.
-Calculate the concentration of the [HC2H3O2] in this solution.
-Calculate the pH of this buffer solution. The Ka for acetic acid is 1.8x10-5.
-If you take half of this solution and add 2 mL of 1.00 M HCl to it, then what is the pH of this new solution?
-If you take half of this solution and add 2 mL of 1.00 M NaOH to it, then what is the pH of this new solution?
Hubert KochLv2
13 Dec 2019