1
answer
0
watching
259
views
12 Dec 2019
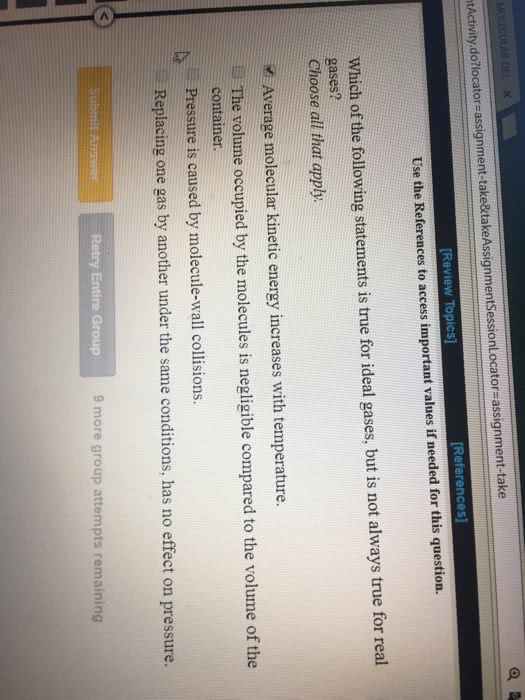
Which of the following is NOT true in the kinetic molecular theory of ideal gases?
The size of the molecules are negligible compared to the volume of the container
The kinetic energy of a sample is proportional to the temperature of the gas.
Collisions are elastic (no friction occurs between the molecules when they collide)
The pressure of a sample is affected by which gases are present.
AND WHY?
Which of the following is NOT true in the kinetic molecular theory of ideal gases?
The size of the molecules are negligible compared to the volume of the container
The kinetic energy of a sample is proportional to the temperature of the gas.
Collisions are elastic (no friction occurs between the molecules when they collide)
The pressure of a sample is affected by which gases are present.
AND WHY?
Sixta KovacekLv2
13 Dec 2019