1
answer
0
watching
507
views
12 Dec 2019
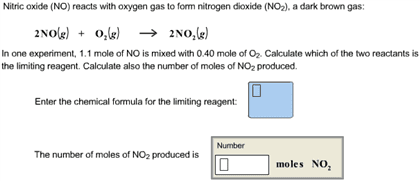
Nitric oxide (NO) reacts with oxygen gas to form nitrogen dioxide (NO2), a dark brown gas:
2NO(g) + O2(g)â 2NO2(g)
In one experiment, 0.857 mol of NO is mixed with 0.498 mol of O2. Determine which of the two reactants is the limiting reactant. Calculate also the number of moles of NO2 produced.
Limiting reactant:
Moles of NO2 produced: _____ moles
Nitric oxide (NO) reacts with oxygen gas to form nitrogen dioxide (NO2), a dark brown gas:
2NO(g) + O2(g)â 2NO2(g)
In one experiment, 0.857 mol of NO is mixed with 0.498 mol of O2. Determine which of the two reactants is the limiting reactant. Calculate also the number of moles of NO2 produced.
Limiting reactant:
Moles of NO2 produced: _____ moles
Liked by demonmothh
Jamar FerryLv2
13 Dec 2019