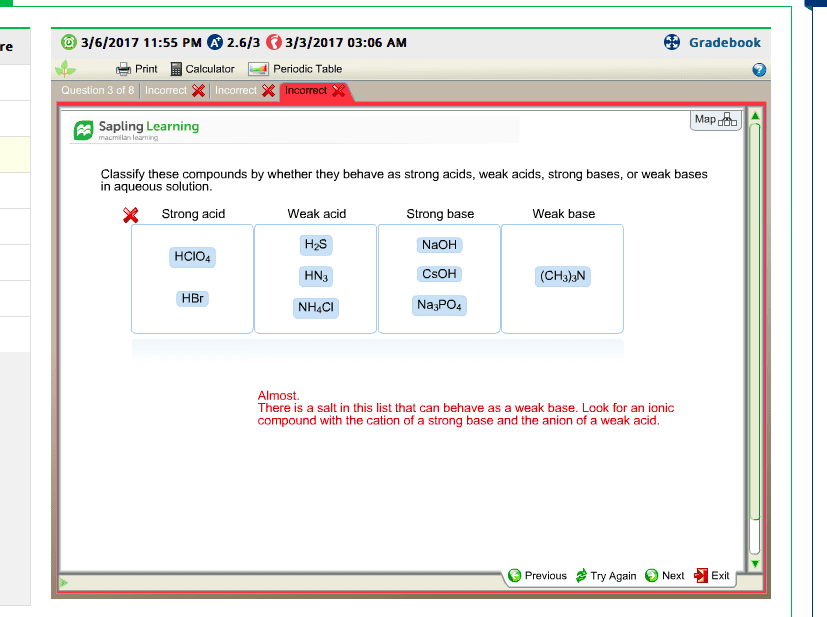
I know the difference between a Strong Acid and a Strong Base.
I understand that all the Strong Acids start with H and are 7 that you have to memorize.
I understand that all the Strong Bases are in the Group 1 and Group 2 of the periodic Table and you just add OH at the end...
My question is that I want to be able to differentiate a weak acid from a weak base. Not about strong acids and strong bases.
I have been making this question to my professor and he says that all the acids that are not in the list of the 7 strong acids are weak and all the bases that are not in the list of the strong bases are weak bases and I know that.
My question is that how can I differentiate a weak acid from a weak base.
This is not about Cojugate base of a weak acid or conjugate acid of a weak base
I want to know if there is a way to know for example if I just have "C5H5N" How can I know if this is a weak base or a weak acid?
Do all the weak bases have something in common that I can see and differentiate them from the weak acids? or do all the weak acids have something in common that I can see and differentiate them from a weak base?
I know the difference between a Strong Acid and a Strong Base.
I understand that all the Strong Acids start with H and are 7 that you have to memorize.
I understand that all the Strong Bases are in the Group 1 and Group 2 of the periodic Table and you just add OH at the end...
My question is that I want to be able to differentiate a weak acid from a weak base. Not about strong acids and strong bases.
I have been making this question to my professor and he says that all the acids that are not in the list of the 7 strong acids are weak and all the bases that are not in the list of the strong bases are weak bases and I know that.
My question is that how can I differentiate a weak acid from a weak base.
This is not about Cojugate base of a weak acid or conjugate acid of a weak base
I want to know if there is a way to know for example if I just have "C5H5N" How can I know if this is a weak base or a weak acid?
Do all the weak bases have something in common that I can see and differentiate them from the weak acids? or do all the weak acids have something in common that I can see and differentiate them from a weak base?