1
answer
0
watching
146
views
12 Dec 2019
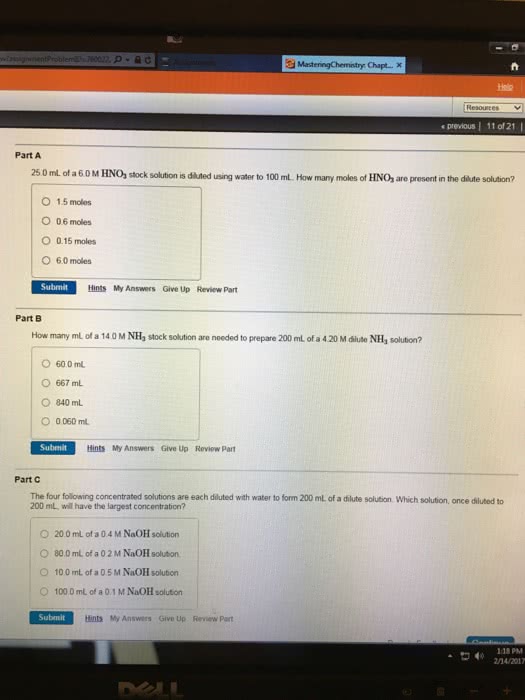
Dilution of a more concentrated sample can be determined using the concept that the number o moless in the samplee qliquote is not changed when water or other solutions are addedde to dilute the sample. If 10.0 mL of 0.00200 M Fe(NO3)3 is diluted to 100.0 mL what is the concentration of Fe+3 ion in the diluted solution?
Dilution of a more concentrated sample can be determined using the concept that the number o moless in the samplee qliquote is not changed when water or other solutions are addedde to dilute the sample. If 10.0 mL of 0.00200 M Fe(NO3)3 is diluted to 100.0 mL what is the concentration of Fe+3 ion in the diluted solution?
Nestor RutherfordLv2
13 Dec 2019