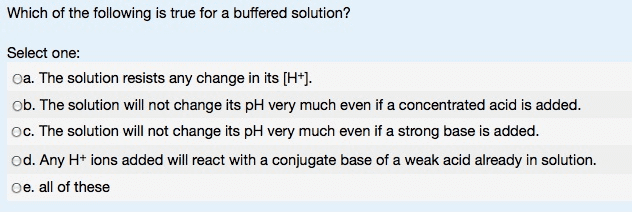
1
answer
0
watching
566
views
12 Dec 2019
A buffer solution is all of the following EXCEPT:
a) a solution that contains both a weak acid and its conjugate base.
b)a solution that regulates pH because it is such a strong acid or base.
c) a solution that resists a change in pH when a base is added.
d) a solution that resists a change in pH when an acid is adde
e) All of the above are true.
A buffer solution is all of the following EXCEPT:
a) a solution that contains both a weak acid and its conjugate base.
b)a solution that regulates pH because it is such a strong acid or base.
c) a solution that resists a change in pH when a base is added.
d) a solution that resists a change in pH when an acid is adde
e) All of the above are true.
Nelly StrackeLv2
13 Dec 2019