0
answers
0
watching
124
views
12 Dec 2019
1) A solution contains 1.03Ã10-2 M nickel(II) nitrate and 9.43Ã10-3 M iron(III) acetate. Solid potassium hydroxide is added slowly to this mixture. What is the concentration of iron(III) ion when nickel ion begins to precipitate?
[Fe3+] = ______M
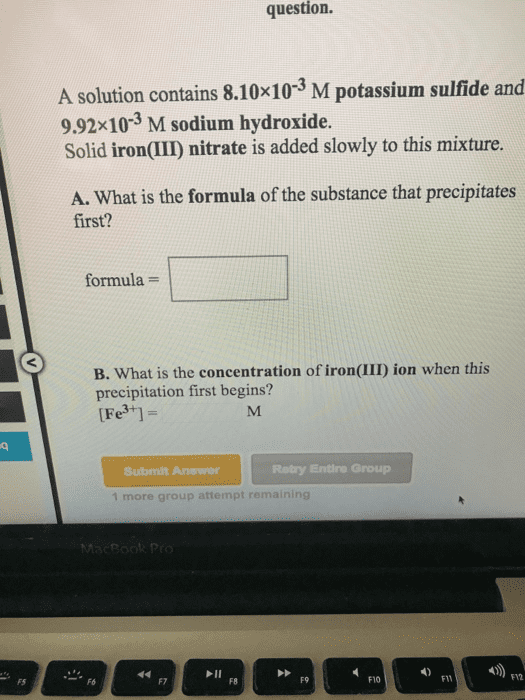
A. What is the formula of the substance that precipitates first?
formula = _______
B. What is the concentration of hydroxide ion when this precipitation first begins?
[OH-] = _____M
1) A solution contains 1.03Ã10-2 M nickel(II) nitrate and 9.43Ã10-3 M iron(III) acetate. Solid potassium hydroxide is added slowly to this mixture. What is the concentration of iron(III) ion when nickel ion begins to precipitate?
[Fe3+] = ______M
A. What is the formula of the substance that precipitates first?
| formula = | _______ |
B. What is the concentration of hydroxide ion when this precipitation first begins?
[OH-] = _____M