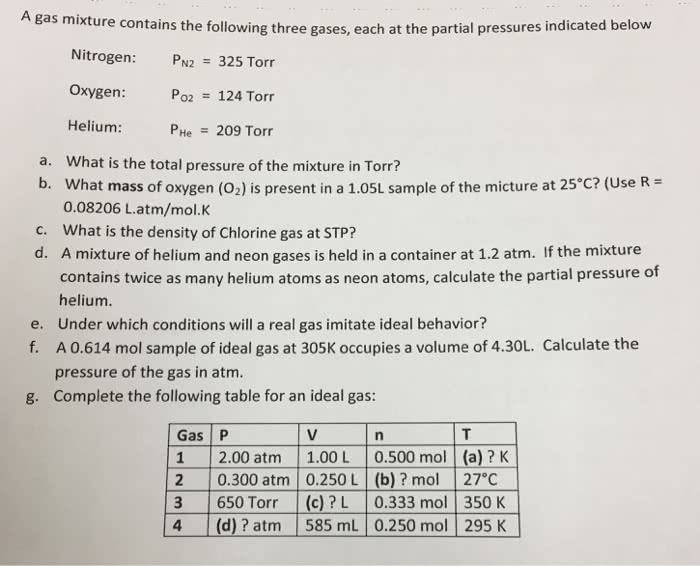
1
answer
1
watching
1,535
views
12 Dec 2019
A mixture of argon and neon has a measured gas density of 1.50 g/L at STP. Assuming ideal-gas behavior, what is the average molar mass of the mixture? What is the percent composition of the mixture?
A mixture of argon and neon has a measured gas density of 1.50 g/L at STP. Assuming ideal-gas behavior, what is the average molar mass of the mixture? What is the percent composition of the mixture?
Jean KeelingLv2
13 Dec 2019