2
answers
0
watching
414
views
27 Sep 2018
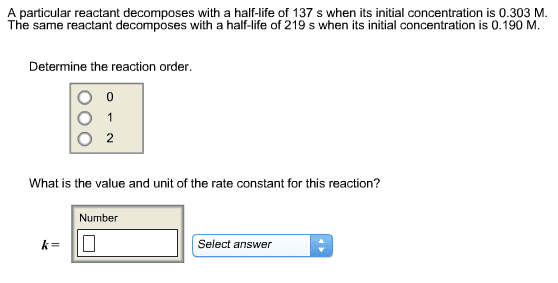
Ni(CO)4 (s) = 4CO (g) + Ni (s)
At 66.0C, the rate constant for this reaction is 1.873 s^-1
a) what is the reaction order for this process
b) Write the rate law expression for this reaction based on youranswer to part a.
c) Determine the time required for half of the Ni(CO)4 todecompose
d) determine the time required for 90% of the Ni(CO)4 todecompose
please show as much work as possible and provide explanation ifpossible, so that i am able to do it on my own in the future. ThankYou.
Ni(CO)4 (s) = 4CO (g) + Ni (s)
At 66.0C, the rate constant for this reaction is 1.873 s^-1
a) what is the reaction order for this process
b) Write the rate law expression for this reaction based on youranswer to part a.
c) Determine the time required for half of the Ni(CO)4 todecompose
d) determine the time required for 90% of the Ni(CO)4 todecompose
please show as much work as possible and provide explanation ifpossible, so that i am able to do it on my own in the future. ThankYou.
2
answers
0
watching
414
views
For unlimited access to Homework Help, a Homework+ subscription is required.
Nelly StrackeLv2
27 Sep 2018
Already have an account? Log in
Related textbook solutions
Basic Chemistry
5 Edition,
Timberlake
ISBN: 9780134138046
Principles of Chemistry Molecular Approach
4th Edition,
Tro
ISBN: 9780134112831
Chemistry: Structure and Properties
2nd Edition,
Tro
ISBN: 9780134293936
Principles of Chemistry Molecular Approach
3rd Edition, 2014
Tro
ISBN: 9780321971944
Chemistry: A Molecular Approach
3rd Edition,
Tro
ISBN: 9780321809247
Chemistry: A Molecular Approach
5th Edition,
Tro
ISBN: 9780134874371
Principles of Chemistry: A Molecular Approach
4th Edition,
Tro
ISBN: 9780134895741
Chemistry: The Central Science
14th Edition, 2017
Brown
ISBN: 9780134414232