1
answer
0
watching
114
views
12 Dec 2019
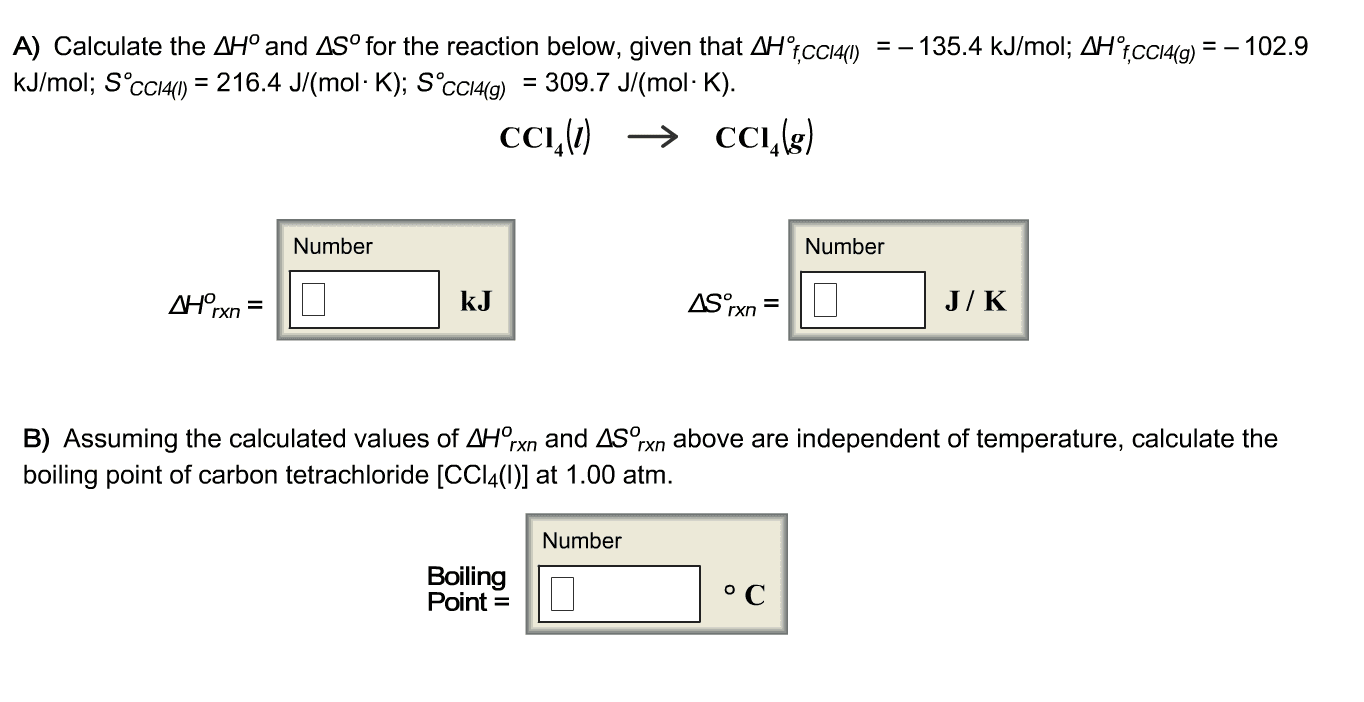
Show what the process CCL4(l)>CCL4(g) favors the ractant (liquid) at 25 degrees C by estimating its normal boiling point ot ther critical Temperature of this process from the following data:
ÎH°fâ, kJ/mol S°f, J/K*mol CCl4(l) -135.4 216.4 CCl4(g) -102.9 309.7
Show what the process CCL4(l)>CCL4(g) favors the ractant (liquid) at 25 degrees C by estimating its normal boiling point ot ther critical Temperature of this process from the following data:
| ÎH°fâ, kJ/mol | S°f, J/K*mol | |
| CCl4(l) | -135.4 | 216.4 |
| CCl4(g) | -102.9 | 309.7 |
Nestor RutherfordLv2
13 Dec 2019