1
answer
0
watching
417
views
12 Dec 2019
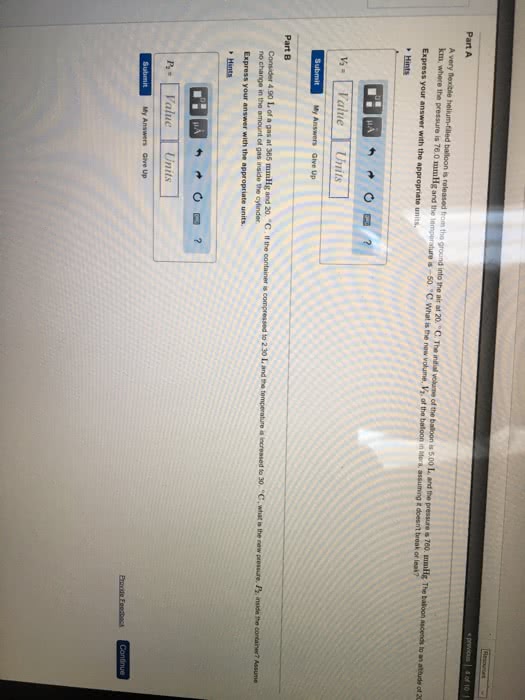
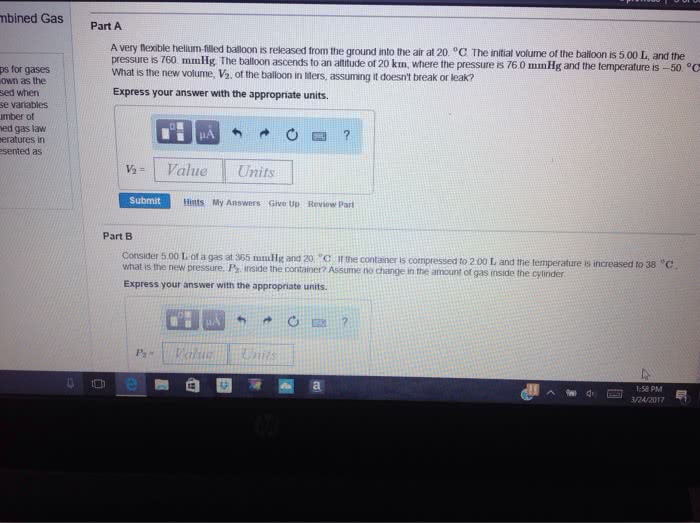
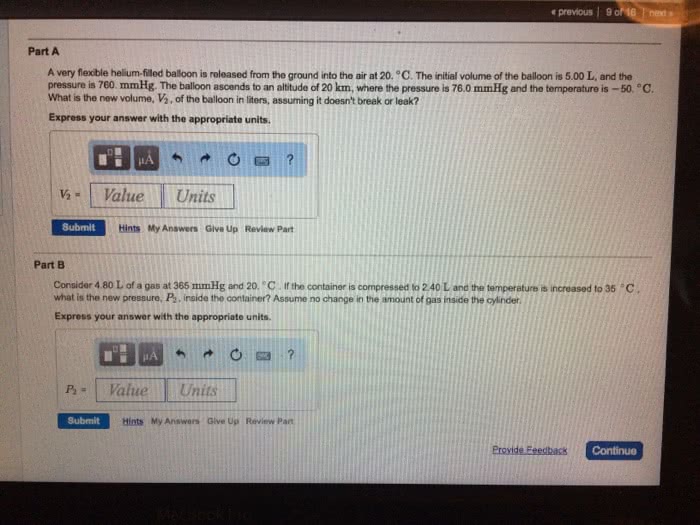
A very flexible helium-filled balloon is released from the ground into the air at 20 *C. The initial volume of the balloon is 5.00 L, and the pressure is 760 mmHg. The balloon ascends to an altitude of 20 km, where the pressure is 76.0 mmHg and the temperature is -50*C. What is the new volume, V2, of the balloon in liters, assuming it doesn't break or leak?
(Express your answer numerically in liters)
A very flexible helium-filled balloon is released from the ground into the air at 20 *C. The initial volume of the balloon is 5.00 L, and the pressure is 760 mmHg. The balloon ascends to an altitude of 20 km, where the pressure is 76.0 mmHg and the temperature is -50*C. What is the new volume, V2, of the balloon in liters, assuming it doesn't break or leak?
(Express your answer numerically in liters)
Bunny GreenfelderLv2
13 Dec 2019