0
answers
0
watching
73
views
13 Dec 2019
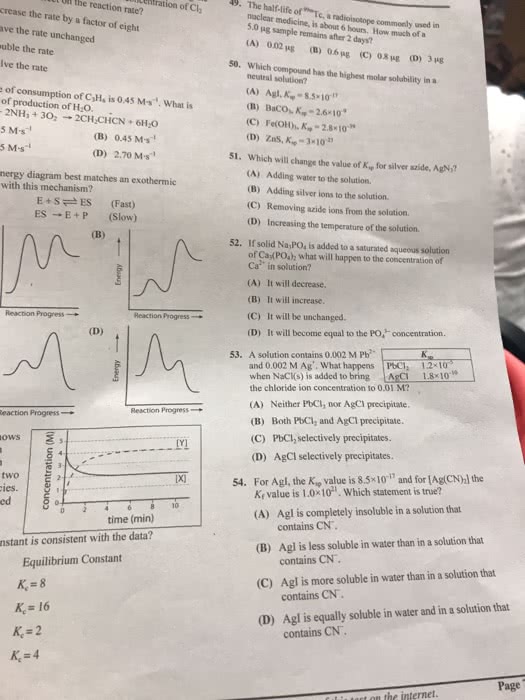
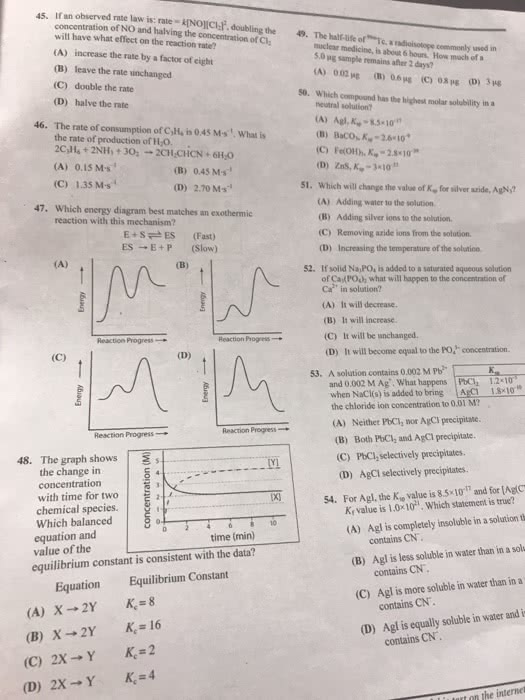
1. The reaction below describes the process of silver phosphate dissolution in water. Ag3(PO4)(s) 3Ag+(aq) + PO43-(aq) What would be the effect of adding HClO4 to a saturated solution represented by the equilibrium reaction shown above? (51%) a. The added HClO4 would have no effect b. The added HClO4 would protonate PO43-(aq), causing Ag+(aq) to precipitate c. The added HClO4 would react with Ag+(aq), forming HAg(aq) and causing more Ag3PO4(s) to dissolve d. The added HClO4 would protonate PO43-(aq), causing more Ag3PO4(s) to dissolve
1. The reaction below describes the process of silver phosphate dissolution in water. Ag3(PO4)(s) 3Ag+(aq) + PO43-(aq) What would be the effect of adding HClO4 to a saturated solution represented by the equilibrium reaction shown above? (51%) a. The added HClO4 would have no effect b. The added HClO4 would protonate PO43-(aq), causing Ag+(aq) to precipitate c. The added HClO4 would react with Ag+(aq), forming HAg(aq) and causing more Ag3PO4(s) to dissolve d. The added HClO4 would protonate PO43-(aq), causing more Ag3PO4(s) to dissolve
0
answers
0
watching
73
views
For unlimited access to Homework Help, a Homework+ subscription is required.
Related textbook solutions
Basic Chemistry
5 Edition,
Timberlake
ISBN: 9780134138046
Principles of Chemistry Molecular Approach
4th Edition,
Tro
ISBN: 9780134112831
Chemistry: Structure and Properties
2nd Edition,
Tro
ISBN: 9780134293936
Principles of Chemistry Molecular Approach
3rd Edition, 2014
Tro
ISBN: 9780321971944
Chemistry: A Molecular Approach
3rd Edition,
Tro
ISBN: 9780321809247
Chemistry: A Molecular Approach
5th Edition,
Tro
ISBN: 9780134874371
Principles of Chemistry: A Molecular Approach
4th Edition,
Tro
ISBN: 9780134895741
Chemistry: The Central Science
14th Edition, 2017
Brown
ISBN: 9780134414232