0
answers
0
watching
92
views
13 Dec 2019
For the titration of 40.0 mL of 0.0100 M Sn2 by 0.0500 M Tl3 in 1 M HCl, using Pt and Ag | AgCl electrodes:
(a) What is the balanced titration reaction?
(b) What are the two half-reactions that occur at the indicator electrode? Answer by adding charges, coefficients, and products to the following.
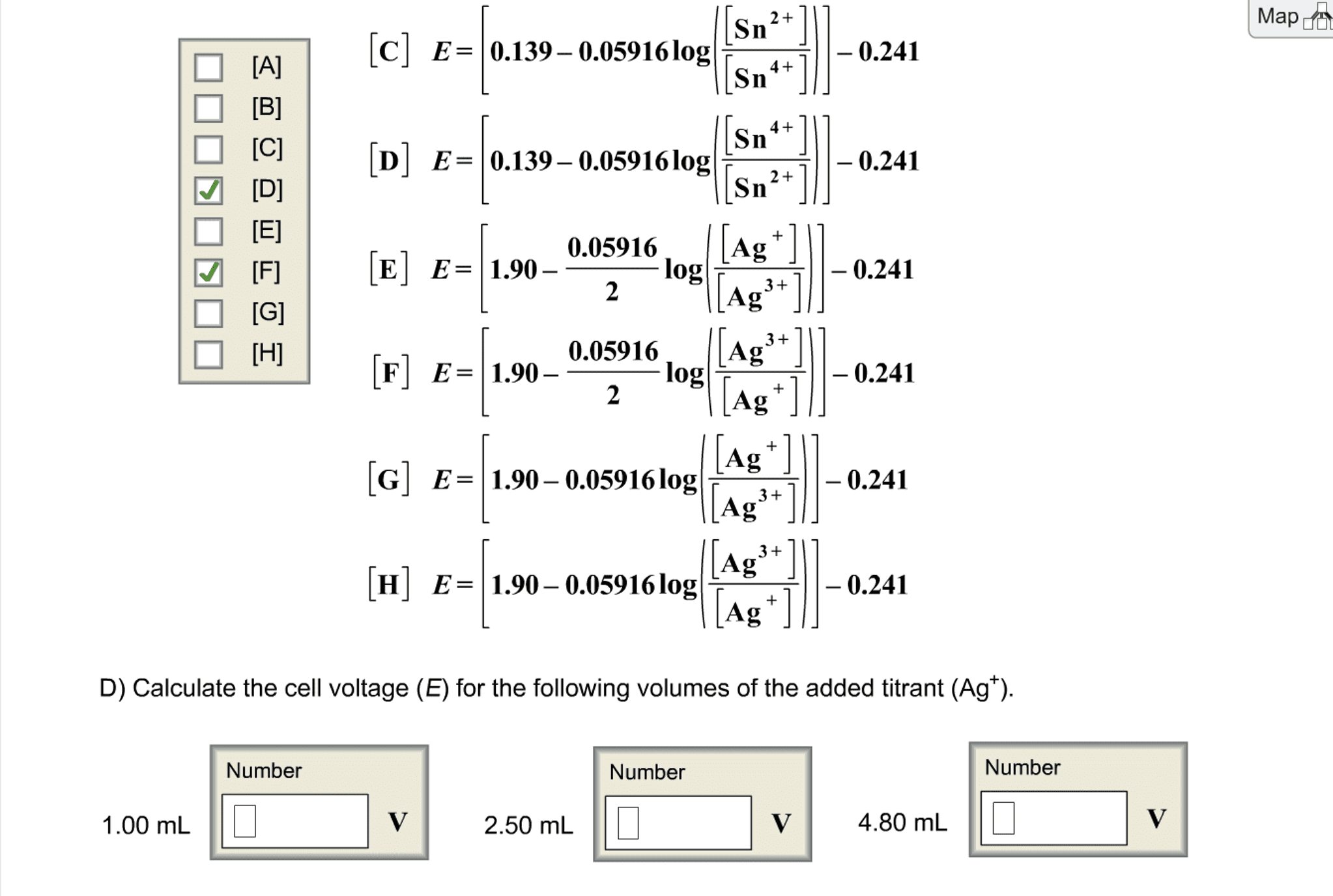
(c) What are the two Nernst equations for the cell voltage? The potential for the Ag | AgCl electrode is 0.197 V.
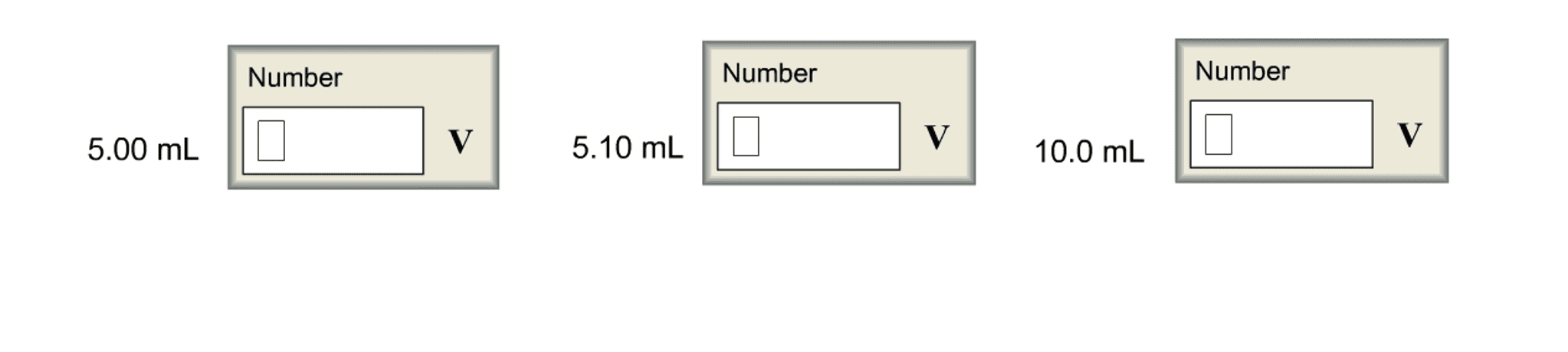
(d) What is the value of E at the following volumes of added Tl3 ?
1.00mL
4.00mL
7.9mL
8.00mL
8.10mL
13.00mL
For the titration of 40.0 mL of 0.0100 M Sn2 by 0.0500 M Tl3 in 1 M HCl, using Pt and Ag | AgCl electrodes:
(a) What is the balanced titration reaction?
(b) What are the two half-reactions that occur at the indicator electrode? Answer by adding charges, coefficients, and products to the following.
(c) What are the two Nernst equations for the cell voltage? The potential for the Ag | AgCl electrode is 0.197 V.
(d) What is the value of E at the following volumes of added Tl3 ?
1.00mL
4.00mL
7.9mL
8.00mL
8.10mL
13.00mL