2
answers
0
watching
218
views
21 Apr 2018
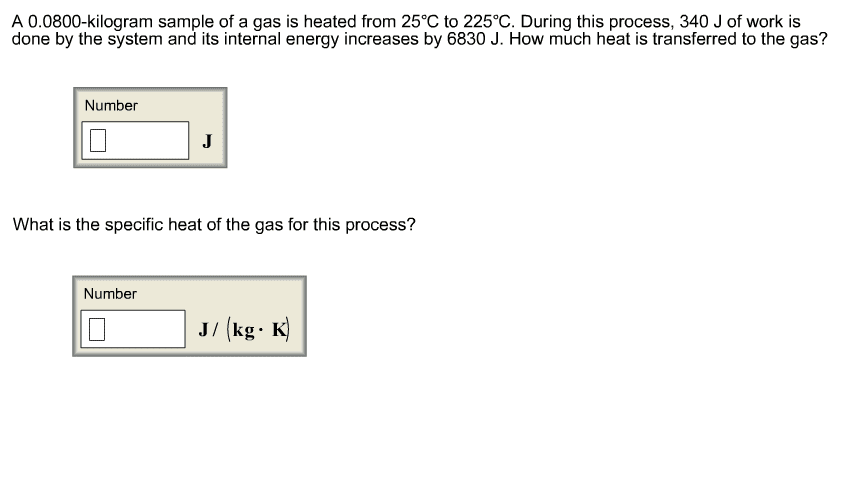
An 80.0-gram sample of gas was heated from 25°C to 225°C. During this process, 346 J of work was done by the system, and its internal energy increased by 8455 J. What is the specific heat of the gas?
An 80.0-gram sample of gas was heated from 25°C to 225°C. During this process, 346 J of work was done by the system, and its internal energy increased by 8455 J. What is the specific heat of the gas?
25 Jun 2023
2 Jun 2021
Already have an account? Log in