1
answer
0
watching
95
views
23 Apr 2019
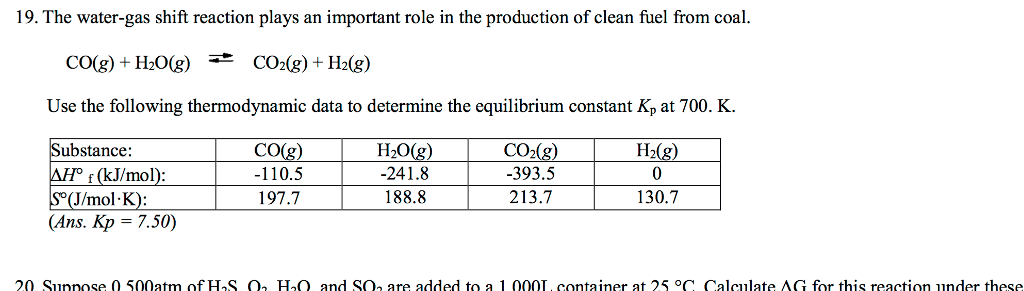
The water-gas shift reaction plays an important role in the production of clean fuel from coal.

After reacting for a period of time at 700K, the reaction mixture contains 



Substance



-110.5
197.7

-241.8
188.8

-393.5
213.7

0
130.7
Is the reaction at equilibrium? If not, in which direction will the reaction proceed to reach equilibrium?
The water-gas shift reaction plays an important role in the production of clean fuel from coal.

After reacting for a period of time at 700K, the reaction mixture contains 



|
Substance |
|
|
|
|
-110.5 |
197.7 |
|
|
-241.8 |
188.8 |
|
|
-393.5 |
213.7 |
|
|
0 |
130.7 |
Is the reaction at equilibrium? If not, in which direction will the reaction proceed to reach equilibrium?
Robert KubaraLv10
18 Feb 2021