At elevated temperatures, CH3NC isomerizes to CH3CN:
CH3NC (g) to CH3 CN (g)
At the start of the experiment, there are .200 mol of reactant (CH3NC) and 0 mol of product (CH3CN) in the reaction vessel. After 25 min of reaction, .108 mol of reactant remains. The average rate of decomposition of CH3NC in this 25 min period is ?
At elevated temperatures, CH3NC isomerizes to CH3CN:
CH3NC (g) to CH3 CN (g)
At the start of the experiment, there are .200 mol of reactant (CH3NC) and 0 mol of product (CH3CN) in the reaction vessel. After 25 min of reaction, .108 mol of reactant remains. The average rate of decomposition of CH3NC in this 25 min period is ?
For unlimited access to Homework Help, a Homework+ subscription is required.
Related textbook solutions
Basic Chemistry
Principles of Chemistry Molecular Approach
Chemistry: Structure and Properties
Principles of Chemistry Molecular Approach
Chemistry: A Molecular Approach
Chemistry: A Molecular Approach
Principles of Chemistry: A Molecular Approach
Chemistry: The Central Science
Related questions
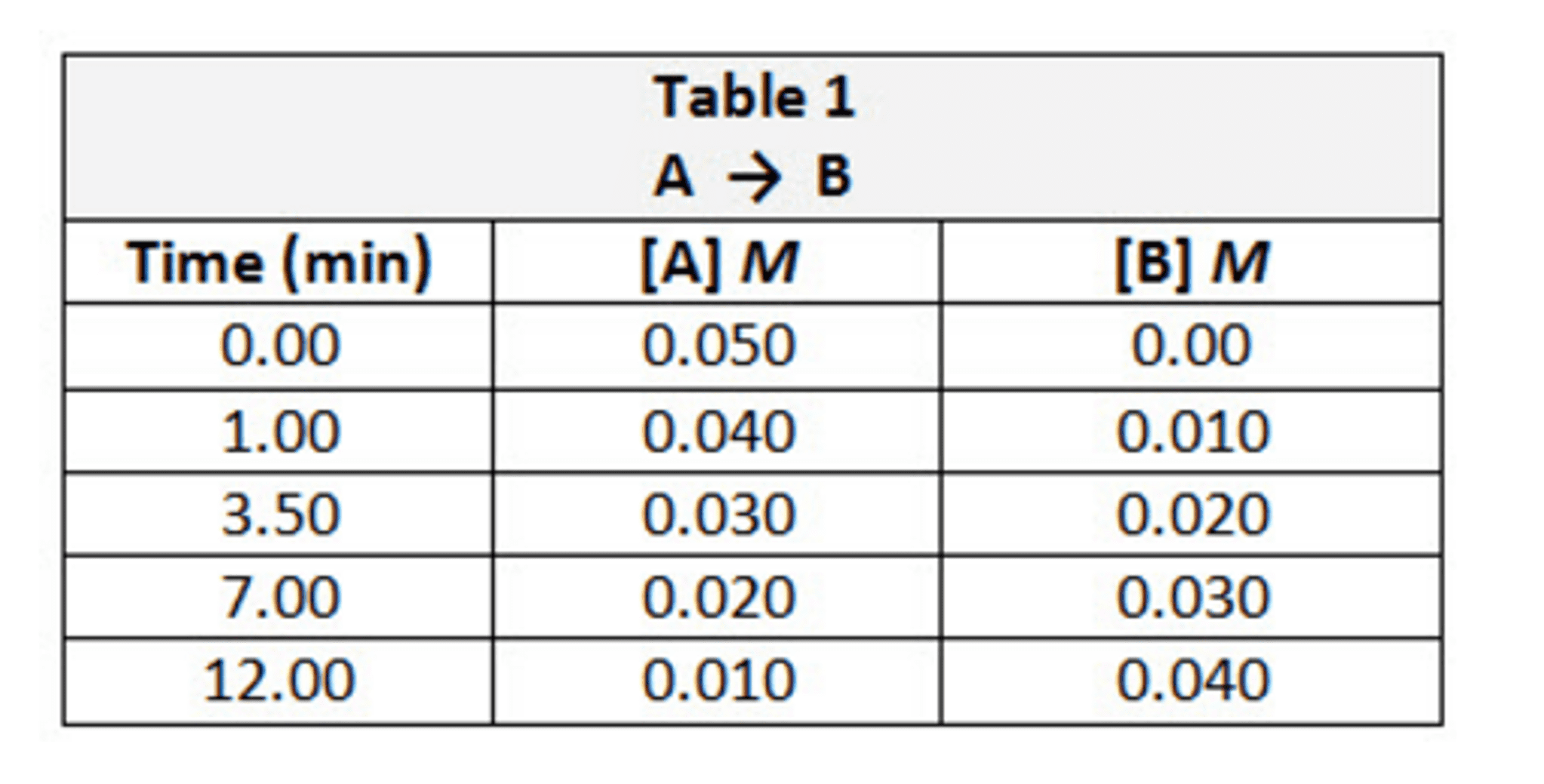
Part B - Use the data in(Figure 1) to calculate reaction rate in terms of B during the time period of 1.00 min to 3.50 min.
Part C
The reaction rates calculated above in terms of A and B during the same time period (1.00 min to 3.50 min) are Please Choosedifferent. the same.
Part D - For the generic reaction: A? B
The Rate Expression is written as: Reaction Rate=?1a?[A]?t=1b?[B]?t
Why is there a negative sign in the expression for reaction rate in terms of the reactant A?
Part E - What does a negative sign in any rate expression mean?
Select all that apply.
| The substance is being consumed. |
| The reaction is proceeding backward. |
| The substance is a reactant. |
| The rate of the reaction is decreasing. |
| None of the above. |
calculate and compare the average rate during different time periods
for the same reaction, A? B
Part F - Calculate the average rate between 3.50 â 7.00 min.
| | |||
| M/min |
Part G - Calculate the average rate between 7.00 â 12.00 min.
| | |||
| M/min |
Part H
The reaction rate Please Choose
increases
decreases
remains constant
shows no trend as the reaction proceeds.
Part I - Suggest an explanation for this trend.

