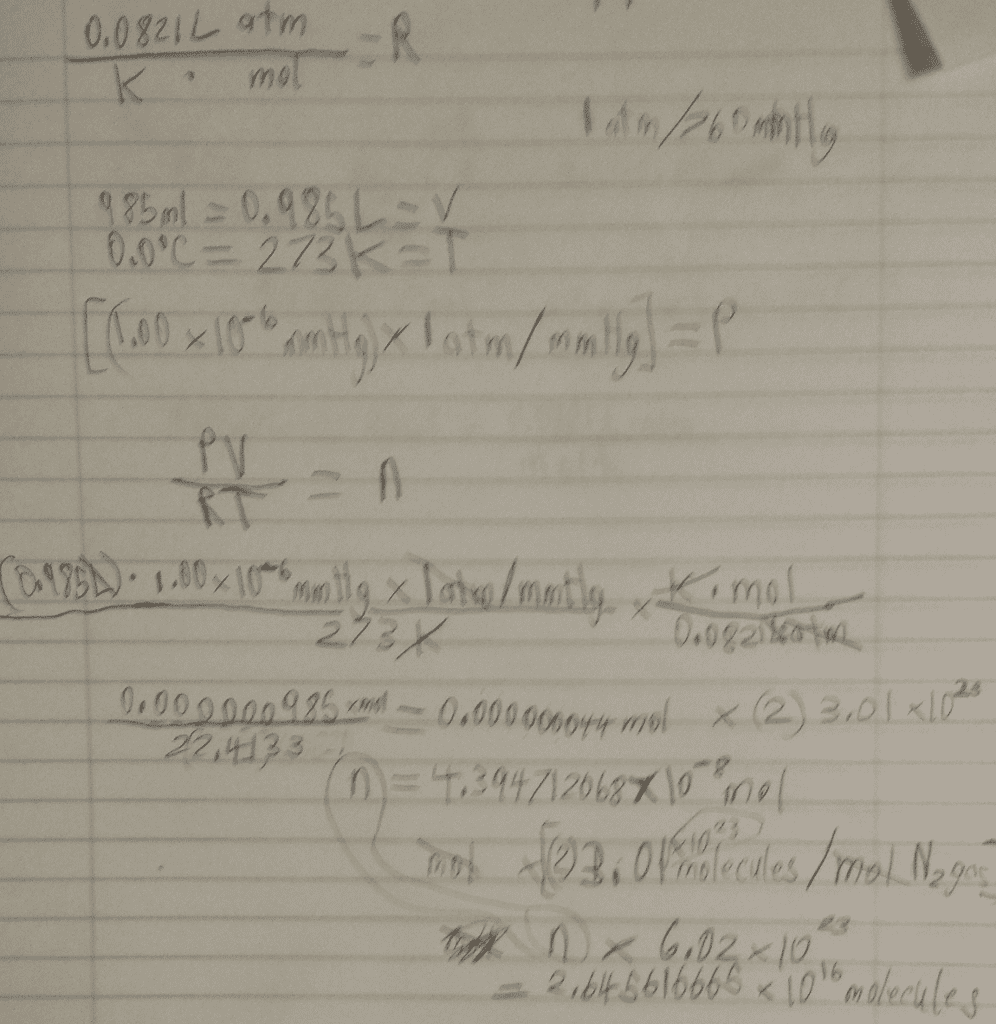1
answer
0
watching
191
views
13 Dec 2019
Did I do this write the answer does not seem right to me
A gas has a volume of 255 mL, a pressure of 733 mm Hg, a temperature of 548 â, and a mass of 1.22g. What is the molar mass?
pV = nRT
convert 733 mm Hg to atm = 733mm Hg x 1 atm/760mm Hg = 0.9645 atm
Convert C to K = 548 â + 273.15 K = 821.15°K
Convert mL to L = 255mL x 1L/1000mL = 0.255L
0.9645 atm x 0.255L= n x 0.8206L*atm/mol*K x 821.15°K
0.2459/ 673.84 = n x 673.84/673.84
.000364 mol = n
M = m/n 1.22g/.000364 = 3351.648
Did I do this write the answer does not seem right to me
A gas has a volume of 255 mL, a pressure of 733 mm Hg, a temperature of 548 â, and a mass of 1.22g. What is the molar mass?
pV = nRT
convert 733 mm Hg to atm = 733mm Hg x 1 atm/760mm Hg = 0.9645 atm
Convert C to K = 548 â + 273.15 K = 821.15°K
Convert mL to L = 255mL x 1L/1000mL = 0.255L
0.9645 atm x 0.255L= n x 0.8206L*atm/mol*K x 821.15°K
0.2459/ 673.84 = n x 673.84/673.84
.000364 mol = n
M = m/n 1.22g/.000364 = 3351.648
Jean KeelingLv2
17 Dec 2019