1
answer
0
watching
76
views
13 Dec 2019
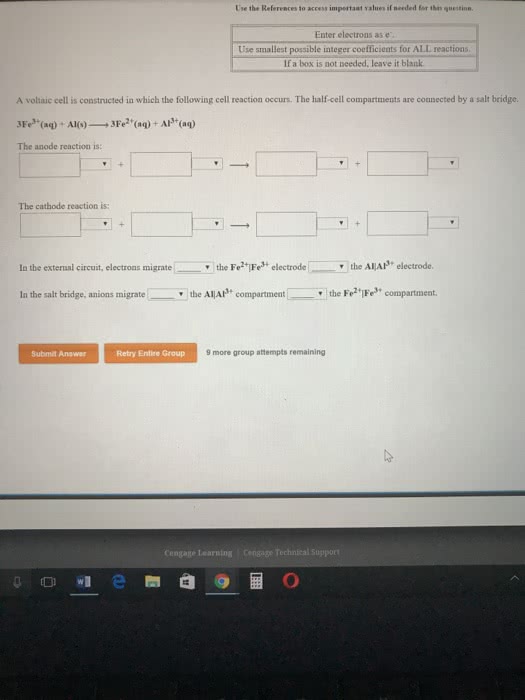
The iron-chromium voltaic cell utilizes the following reactionwith a measured potential of 0.30V. What is the standard change inG for this reaction. Please show all work.
3Fe^2+(aq) + 2Cr(s) --> 3Fe(s) + 2Cr^3+(aq)
The iron-chromium voltaic cell utilizes the following reactionwith a measured potential of 0.30V. What is the standard change inG for this reaction. Please show all work.
3Fe^2+(aq) + 2Cr(s) --> 3Fe(s) + 2Cr^3+(aq)
Collen VonLv2
17 Dec 2019