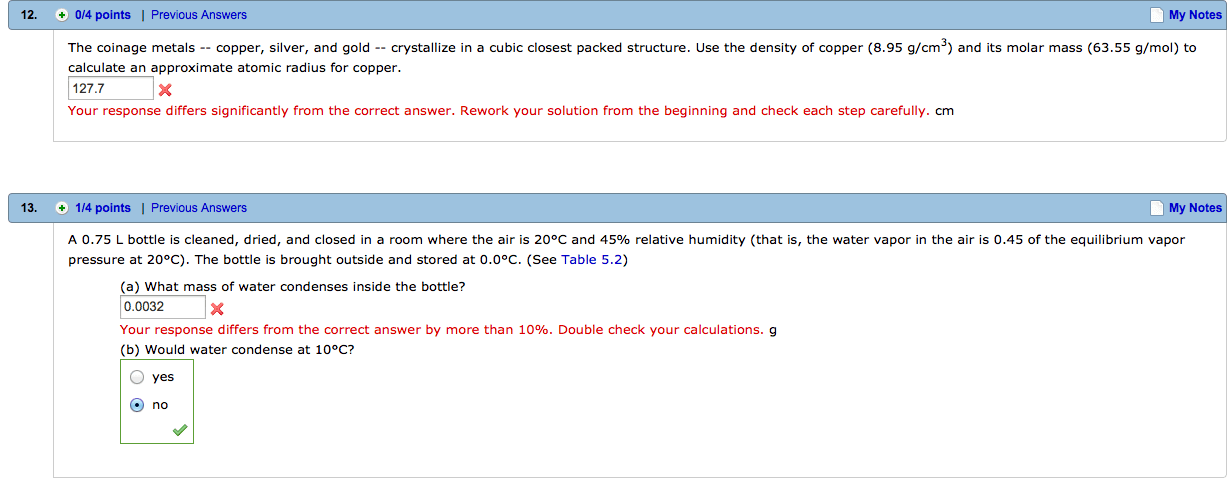
a) From the Chapter 1 quiz: The densities of the coinage metals (copper, silver and gold) are as follows: copper = 8.95 g/cm3, silver = 12.59 g/cm3, gold = 19.32 g/cm3 A sample of material is found to weigh 10.8 grams, and have a volume of 1.20 cm. The sample could be which of the coinage metals? Correct answer: Copper. The most commonly selected incorrect answer on this question was ânone of the aboveâ. Prove that the answer is, indeed, copper and provide an explanation why a student might have chosen ânone of the above.â
b) From the Chapter 5 quiz: âWhich of the following will occur if the temperature of a gas is increased from 20°C to 40°C at constant pressure?â More than ¼ of respondents selected - The volume will double. Explain why this answer is incorrect.
c) From the Chapter 6 quiz: This was the HARDEST question asked all term: âWhich of the following solutions has the highest osmolarity?â The options were: 0.20 M NaCl, 0.15 M BaCl2, 0.10 M AlCl3 and 0.35 M sucrose. The correct answer is 0.15 M BaCl2. The most often selected incorrect response was 0.35 M sucrose. I can now tell you that the BaCl2 has the highest osmolarity and the sucrose is the lowest. Your challenge now is to explain which has the higher osmolarity between the NaCl and the AlCl3. Explain your answer.
d) From the first Chapter 8 quiz: âWhen solid sodium hydroxide is dissolved in water which of the following species is not present in the solution? The correct answer is NaOH(aq). Why is this answer correct?
a) From the Chapter 1 quiz: The densities of the coinage metals (copper, silver and gold) are as follows: copper = 8.95 g/cm3, silver = 12.59 g/cm3, gold = 19.32 g/cm3 A sample of material is found to weigh 10.8 grams, and have a volume of 1.20 cm. The sample could be which of the coinage metals? Correct answer: Copper. The most commonly selected incorrect answer on this question was ânone of the aboveâ. Prove that the answer is, indeed, copper and provide an explanation why a student might have chosen ânone of the above.â
b) From the Chapter 5 quiz: âWhich of the following will occur if the temperature of a gas is increased from 20°C to 40°C at constant pressure?â More than ¼ of respondents selected - The volume will double. Explain why this answer is incorrect.
c) From the Chapter 6 quiz: This was the HARDEST question asked all term: âWhich of the following solutions has the highest osmolarity?â The options were: 0.20 M NaCl, 0.15 M BaCl2, 0.10 M AlCl3 and 0.35 M sucrose. The correct answer is 0.15 M BaCl2. The most often selected incorrect response was 0.35 M sucrose. I can now tell you that the BaCl2 has the highest osmolarity and the sucrose is the lowest. Your challenge now is to explain which has the higher osmolarity between the NaCl and the AlCl3. Explain your answer.
d) From the first Chapter 8 quiz: âWhen solid sodium hydroxide is dissolved in water which of the following species is not present in the solution? The correct answer is NaOH(aq). Why is this answer correct?