1
answer
0
watching
305
views
13 Dec 2019
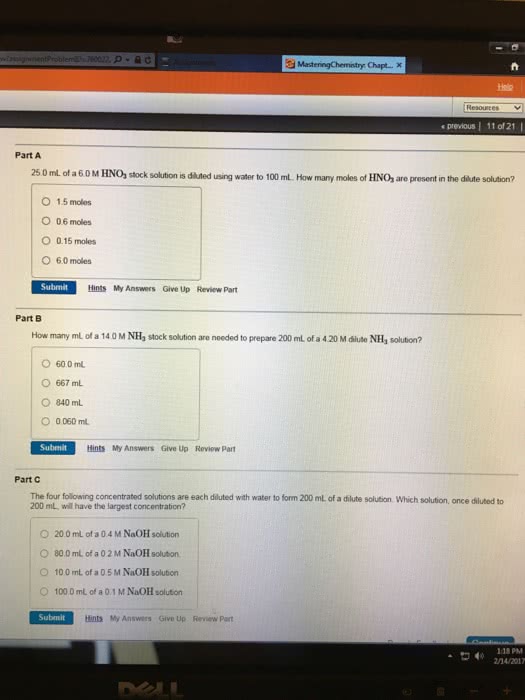
Calculate the final concentration of the solution when water is added to prepare each of the following solutions.
17.0 mL of a 15.0 %(m/v) K2SO4 solution is diluted to 80.0 mL. Answer should be in %(m/v)
80.0 mL of a 6.00 M NaOH solution is diluted to 235 mL. Answer should be in M
Calculate the final concentration of the solution when water is added to prepare each of the following solutions.
17.0 mL of a 15.0 %(m/v) K2SO4 solution is diluted to 80.0 mL. Answer should be in %(m/v)
80.0 mL of a 6.00 M NaOH solution is diluted to 235 mL. Answer should be in M
Tod ThielLv2
17 Dec 2019