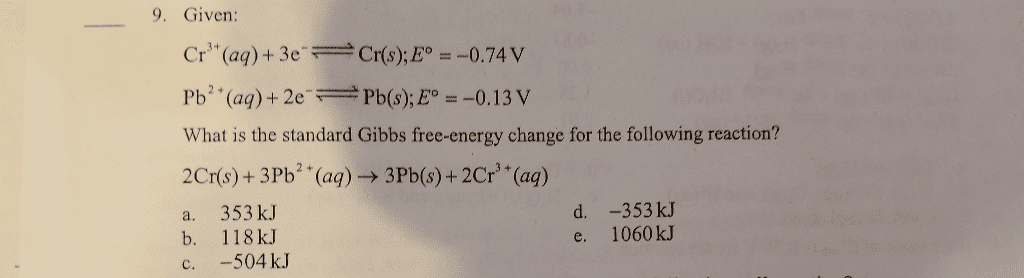1. Calculate the standard change in free energy ?G? (kJ) at 298 K for the reaction C2H4(G)+3o2(G) --> 2CO2(G)+2H2O(G)
a -1310 b. +1180 c. -691 d. +1310 e. -1180
2. The value of E?cell for the reaction 2Cr3+(aq)+6Hg(l) --> 2Cr(s)+3Hg22+(aq) is 1.59 V. Calculate the ?G? (kJ) for the reaction.
a. -921 b. -153 c. -767 d. -460 e. -307
3. What is the value of the equilibrium constant for the cell reaction below at 25 deg C? E?cellÂ=0.61V 2Cr(s)+3Pb2+(aq) <---> 3Pb(s)+2Cr3+(aq)
a. 8.2*10^30 b. 6.7*10^61 c. 9.9*10^99 d. 4.1*10^20 e. 3.3*10^51
4. Which one of the following is not a redox reaction?
a. Na6FeCl8(s)+2Na(l) ---> 8NaCl(s)+Fe(s)
b. Al(OH)3 (aq)+3H+ ---> Al3+(aq)+3H2O(l)
c. CuCl2(aq)+Ni(s) ---> Cu(s)+NiCl2(aq)
d. C6H12O6(s)+6O2(g) ---> 6CO2+6H2O(l)
e. COÂ2(g)+H2(g) ---> CO(g)+ H2O(g)ÂÂÂÂÂÂÂÂ
5. A voltaic cell consists of a Zn/ZN2+ electrode and a Fe/Fe2+ electrode. If [ZN2+] = 0.005M and if [Fe2+] = .0500 M , what is Ecell?
a. .76 b. 0.25 c. 0.45 d. 0.31 e. 0.37
6. The formation constant for the reaction Ag+ (aq)+2NH3(aq) <----> Ag(NH3)2+(aq) is K=1.7*107 at 25 deg C. What is the ?G?(kJ/mol) at this temperature?
a. -23 b. -18 c. -3.5 d. -1.5 e. -41
7. Nitric oxide reacts with chlorine to form NOCL. What is the free energy (kJ) of the reaction at 277 deg C?
a. +144 b.. -41.0 c. -10.3 d. -22.2 e. +41.0
8. Estimate the boiling point (deg C) of phosphoric Acid.
H3PO4(s) <----> HÂ3PO4(l)
?H?f kJ/mol -1279 - -127.17
?G?f kJ/mol -1119 - 1123.6
?S?f J/(k mol) 110.5 - 150.8
a. 42 b. 181 c. 315 d. -92 e. 305
9. Calculate the equilibrium constant, K, for the following reaction at 25?C:
CH4(g)+2H2O(g) <---> Ã CO2(g)+4H2(g) ?G?= -113.6 kJ (please show ICE chart and explain process)
a. 8.2x1019 b. 0.58 c. 0.96 d. 1.4x10-46 e. 1.2x10-20
10. Which of the following results in a decrease in the entropy of the system?
a. O2(g), 300K --> O2(g), 400K b. H2O(s) --> H2O(l) c. N2(g) --> N2(aq) d. NH3(l) --> NH3(g)
e. 2H2O2(g) ---> 2H2O(g)+O2(g)
1. Calculate the standard change in free energy ?G? (kJ) at 298 K for the reaction C2H4(G)+3o2(G) --> 2CO2(G)+2H2O(G)
a -1310 b. +1180 c. -691 d. +1310 e. -1180
2. The value of E?cell for the reaction 2Cr3+(aq)+6Hg(l) --> 2Cr(s)+3Hg22+(aq) is 1.59 V. Calculate the ?G? (kJ) for the reaction.
a. -921 b. -153 c. -767 d. -460 e. -307
3. What is the value of the equilibrium constant for the cell reaction below at 25 deg C? E?cellÂ=0.61V 2Cr(s)+3Pb2+(aq) <---> 3Pb(s)+2Cr3+(aq)
a. 8.2*10^30 b. 6.7*10^61 c. 9.9*10^99 d. 4.1*10^20 e. 3.3*10^51
4. Which one of the following is not a redox reaction?
a. Na6FeCl8(s)+2Na(l) ---> 8NaCl(s)+Fe(s)
b. Al(OH)3 (aq)+3H+ ---> Al3+(aq)+3H2O(l)
c. CuCl2(aq)+Ni(s) ---> Cu(s)+NiCl2(aq)
d. C6H12O6(s)+6O2(g) ---> 6CO2+6H2O(l)
e. COÂ2(g)+H2(g) ---> CO(g)+ H2O(g)ÂÂÂÂÂÂÂÂ
5. A voltaic cell consists of a Zn/ZN2+ electrode and a Fe/Fe2+ electrode. If [ZN2+] = 0.005M and if [Fe2+] = .0500 M , what is Ecell?
a. .76 b. 0.25 c. 0.45 d. 0.31 e. 0.37
6. The formation constant for the reaction Ag+ (aq)+2NH3(aq) <----> Ag(NH3)2+(aq) is K=1.7*107 at 25 deg C. What is the ?G?(kJ/mol) at this temperature?
a. -23 b. -18 c. -3.5 d. -1.5 e. -41
7. Nitric oxide reacts with chlorine to form NOCL. What is the free energy (kJ) of the reaction at 277 deg C?
a. +144 b.. -41.0 c. -10.3 d. -22.2 e. +41.0
8. Estimate the boiling point (deg C) of phosphoric Acid.
H3PO4(s) <----> HÂ3PO4(l)
?H?f kJ/mol -1279 - -127.17
?G?f kJ/mol -1119 - 1123.6
?S?f J/(k mol) 110.5 - 150.8
a. 42 b. 181 c. 315 d. -92 e. 305
9. Calculate the equilibrium constant, K, for the following reaction at 25?C:
CH4(g)+2H2O(g) <---> Ã CO2(g)+4H2(g) ?G?= -113.6 kJ (please show ICE chart and explain process)
a. 8.2x1019 b. 0.58 c. 0.96 d. 1.4x10-46 e. 1.2x10-20
10. Which of the following results in a decrease in the entropy of the system?
a. O2(g), 300K --> O2(g), 400K b. H2O(s) --> H2O(l) c. N2(g) --> N2(aq) d. NH3(l) --> NH3(g)
e. 2H2O2(g) ---> 2H2O(g)+O2(g)