0
answers
0
watching
127
views
13 Dec 2019
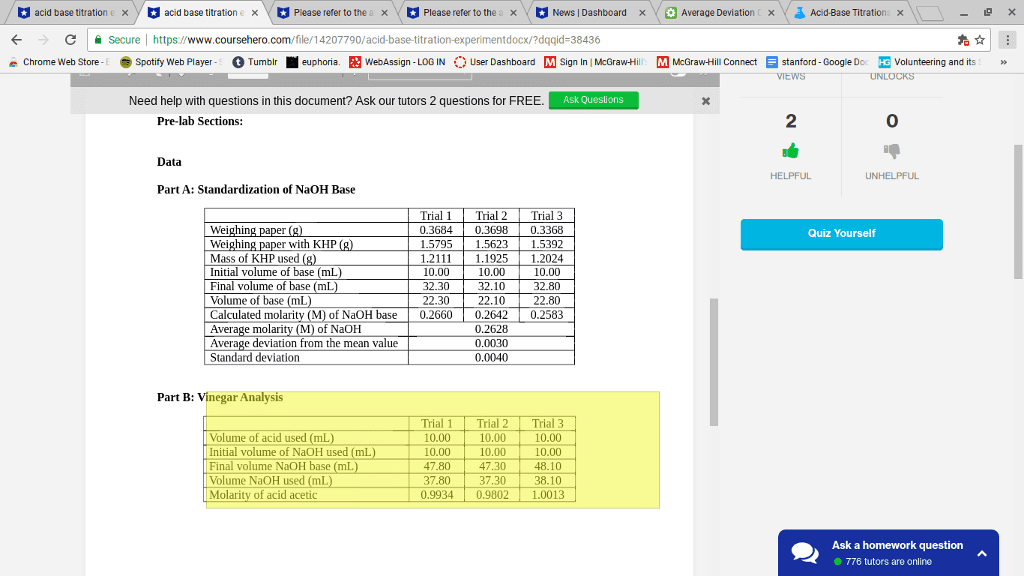
Molarity of standard NaOH solution: x/1 H 2/3 Unknown + 2/3 NaOH -> 2/3 H2O +Na 2/3 Acetate
Vinegar: Titration 1 Titration 2 Tirtration 3 Initial V of Acid .01 mL 5.56 mL 11.32 mL Final V of Acid 5.56 mL 10.71 mL 16.02 mL Inital V of Base .01 mL 27.12 mL 3.01 mL Final V of Base 22.23 mL 44.71 mL 24.90 mL
Unknown Acid code and number of reactive hydrogens: H+3
Unknown Acid Titration 1 Titration 2 (Error) Titration 3 Titration 4 Mass of Acid .1024 g .1028g .1021g .1023g Initial V of Base 24.90 mL 37.80 mL 45.5 mL 9.24 mL Final V of Base 37.80 mL 45.5 mL 9.24 mL 17.80 mL
7. Assuming that the density of vinegar is the same as the density of water, find the mass of vinegar for each titration, and calculate the mass percent of acetic acid forthat titration. (You looked up the density of water back in the measurement lab.)
8. Average your three mass percents of acetic acid to get your final answer for the
mass percent of acetic acid.
9. Find the volumes of base solution for each of your unknown acid titrations.
Molarity of standard NaOH solution: x/1 H 2/3 Unknown + 2/3 NaOH -> 2/3 H2O +Na 2/3 Acetate
| Vinegar: | Titration 1 | Titration 2 | Tirtration 3 |
| Initial V of Acid | .01 mL | 5.56 mL | 11.32 mL |
| Final V of Acid | 5.56 mL | 10.71 mL | 16.02 mL |
| Inital V of Base | .01 mL | 27.12 mL | 3.01 mL |
| Final V of Base | 22.23 mL | 44.71 mL | 24.90 mL |
Unknown Acid code and number of reactive hydrogens: H+3
| Unknown Acid | Titration 1 | Titration 2 (Error) | Titration 3 | Titration 4 |
| Mass of Acid | .1024 g | .1028g | .1021g | .1023g |
| Initial V of Base | 24.90 mL | 37.80 mL | 45.5 mL | 9.24 mL |
| Final V of Base | 37.80 mL | 45.5 mL | 9.24 mL | 17.80 mL |
7. Assuming that the density of vinegar is the same as the density of water, find the mass of vinegar for each titration, and calculate the mass percent of acetic acid forthat titration. (You looked up the density of water back in the measurement lab.)
8. Average your three mass percents of acetic acid to get your final answer for the
mass percent of acetic acid.
9. Find the volumes of base solution for each of your unknown acid titrations.