1
answer
0
watching
597
views
13 Dec 2019
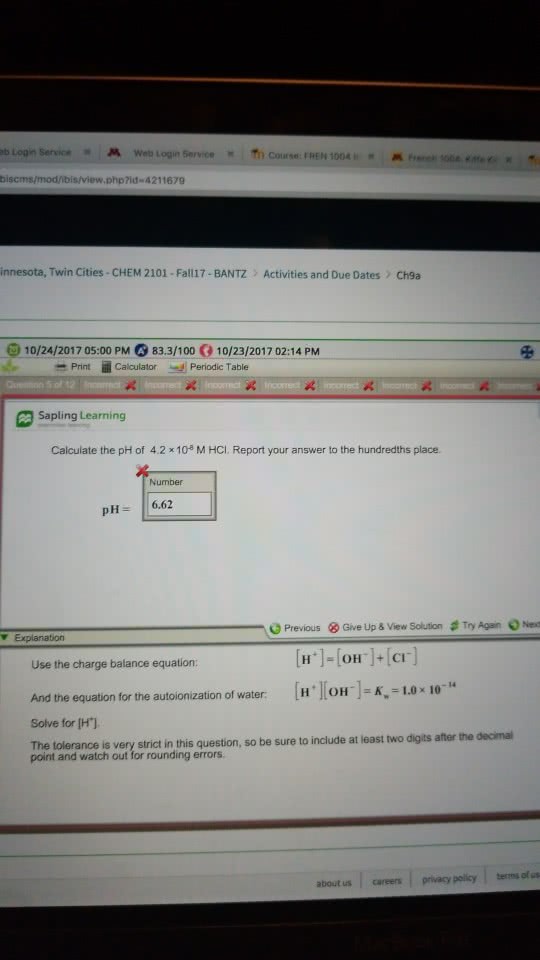
Calculate the pH of 5.8 Ã 10^-8 M HCl. Report your answer to the hundredths place.
What fraction of the total H+ in this solution is from the HCl? Report your answer to the hundredths place.
Hint:
Remember there are H+ ions from the water as well. Use the charge balance equation in the calculation. The tolerance is very strict in this question, so be sure to include at least two digits after the decimal point and watch out for rounding errors.
Calculate the pH of 5.8 Ã 10^-8 M HCl. Report your answer to the hundredths place.
What fraction of the total H+ in this solution is from the HCl? Report your answer to the hundredths place.
Hint:
Remember there are H+ ions from the water as well. Use the charge balance equation in the calculation. The tolerance is very strict in this question, so be sure to include at least two digits after the decimal point and watch out for rounding errors.
Beverley SmithLv2
17 Dec 2019