1
answer
0
watching
262
views
13 Dec 2019
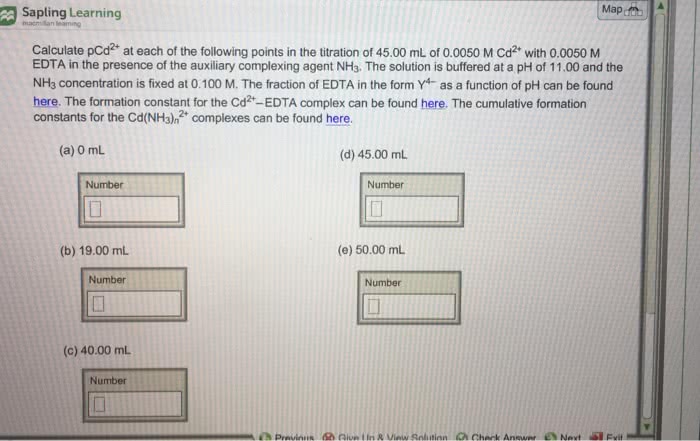
Calculate pCd2 at each of the following points in the titration of 50.00 mL of 0.0040 M Cd2 with 0.0040 M EDTA in the presence of the auxiliary complexing agent NH3. The solution is buffered at a pH of 11.00. Take the NH3 concentration to be a constant 0.100 M. The formation constant for the Cd2 -EDTA complex is given by log Kf = 16.5.
1. 0mL
2. 3.00mL
3. 45mL
4. 50 mL
5. 55mL
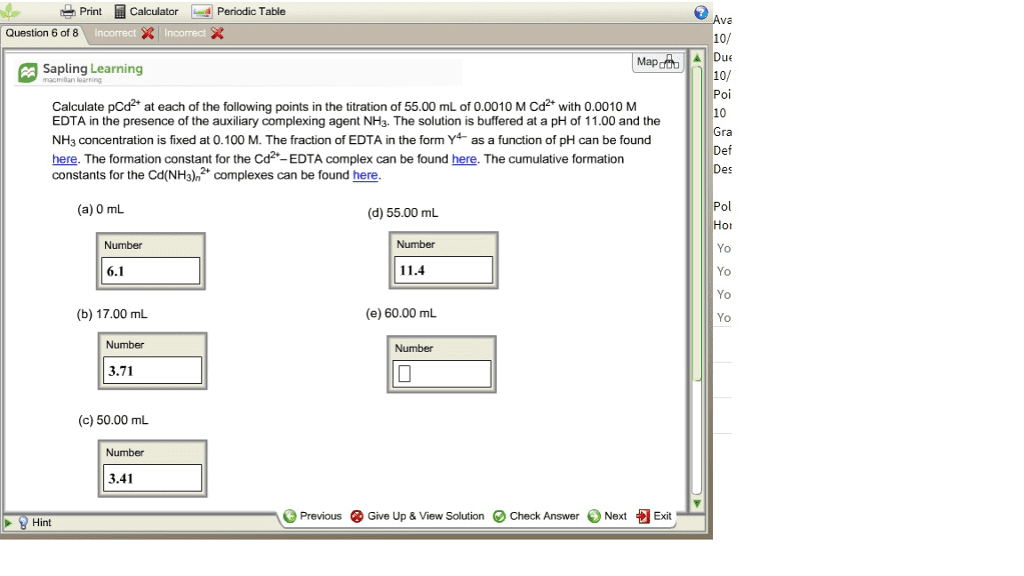
Calculate pCd2 at each of the following points in the titration of 50.00 mL of 0.0040 M Cd2 with 0.0040 M EDTA in the presence of the auxiliary complexing agent NH3. The solution is buffered at a pH of 11.00. Take the NH3 concentration to be a constant 0.100 M. The formation constant for the Cd2 -EDTA complex is given by log Kf = 16.5.
1. 0mL
2. 3.00mL
3. 45mL
4. 50 mL
5. 55mL
Patrina SchowalterLv2
17 Dec 2019