1
answer
0
watching
139
views
13 Dec 2019
\
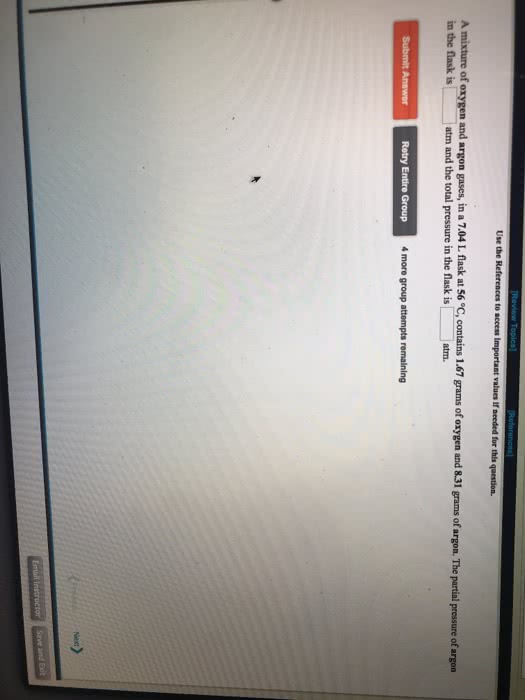
A 1.00 L flask is filled with 1.40 g of argon at 25 âC. A sample of ethane vapor is added to the same flask until the total pressure is 1.050 atm .
What is the partial pressure of argon, PAr, in the flask?
What is the partial pressure of ethane, Pethane, in the flask?
\
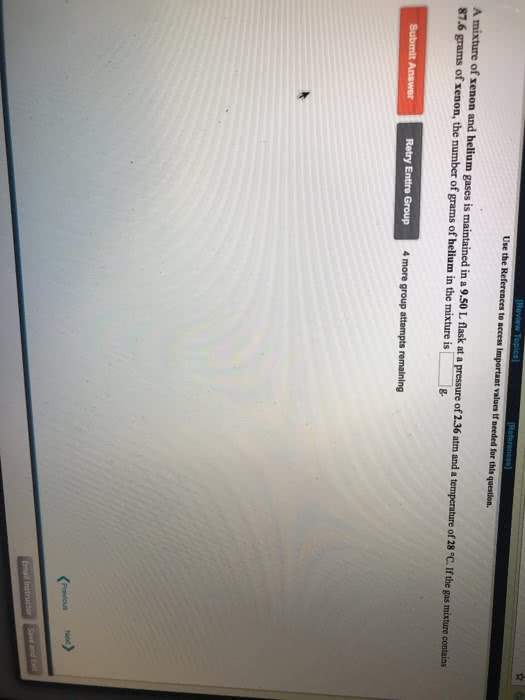
A 1.00 L flask is filled with 1.40 g of argon at 25 âC. A sample of ethane vapor is added to the same flask until the total pressure is 1.050 atm .
What is the partial pressure of argon, PAr, in the flask?
What is the partial pressure of ethane, Pethane, in the flask?
Hubert KochLv2
17 Dec 2019