1
answer
0
watching
212
views
13 Dec 2019
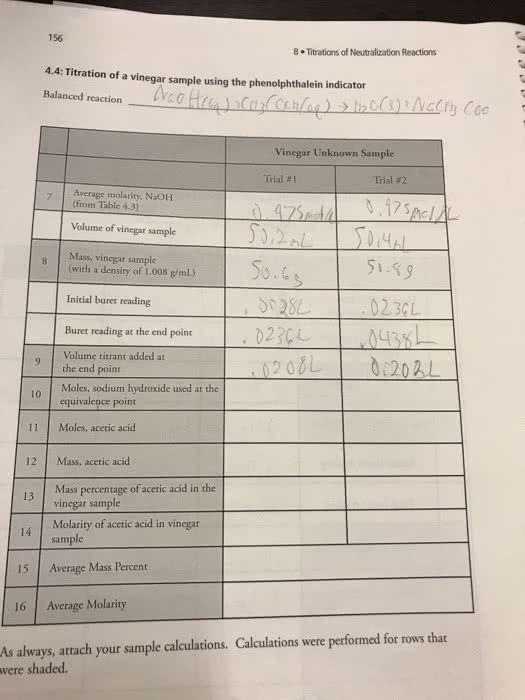
Molarity of NaOH: 0.1388 M
Unknown Acid Buret:
Trial 1
Trial 2
Initial Buret Reading
27.11 ml
17.91 ml
Final Buret Reading
47.31 ml
42.01 ml
Volume of acid added
20.10 ml
24.10 ml
Results:
Trial 1
Trial 2
Volume of NaOH at equivalence point
4.75 mL
14.03 mL
Volume of NaOH at one-half the equivalence point
2.37 mL
7.015 mL
pH at half equivalence point
4.88
4.21
pKa of unknown acid
Average pKa
Average Ka
Moles of unknown acid
Moles
Moles
M of unknown acid
M
M
Average M of unknown acid
can you show the steps as to how to find the answers for the missing boxes, work included as I would like to learn? and a few sentences explaining the results.
Molarity of NaOH: 0.1388 M
Unknown Acid Buret:
| Trial 1 | Trial 2 | |
| Initial Buret Reading | 27.11 ml | 17.91 ml |
| Final Buret Reading | 47.31 ml | 42.01 ml |
| Volume of acid added | 20.10 ml | 24.10 ml |
Results:
| Trial 1 | Trial 2 | |
| Volume of NaOH at equivalence point | 4.75 mL | 14.03 mL |
| Volume of NaOH at one-half the equivalence point | 2.37 mL | 7.015 mL |
| pH at half equivalence point | 4.88 | 4.21 |
| pKa of unknown acid | ||
| Average pKa | ||
| Average Ka | ||
| Moles of unknown acid | Moles | Moles |
| M of unknown acid | M | M |
| Average M of unknown acid |
can you show the steps as to how to find the answers for the missing boxes, work included as I would like to learn? and a few sentences explaining the results.
Reid WolffLv2
17 Dec 2019