0
answers
0
watching
84
views
13 Dec 2019
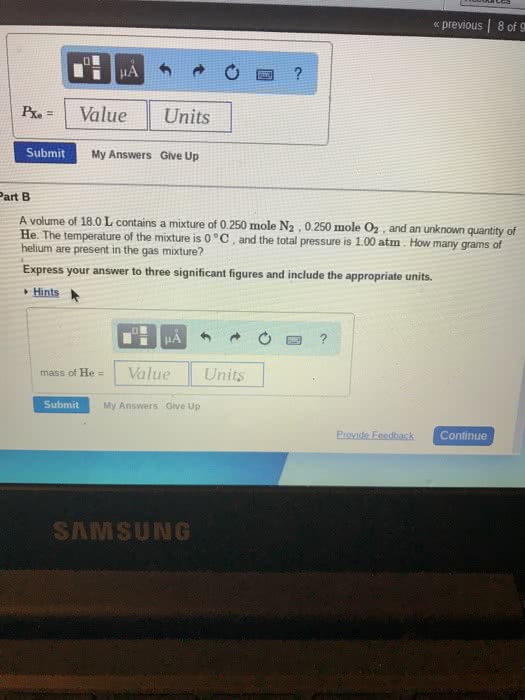
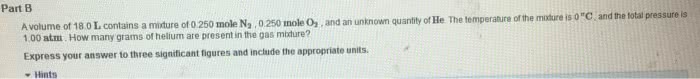
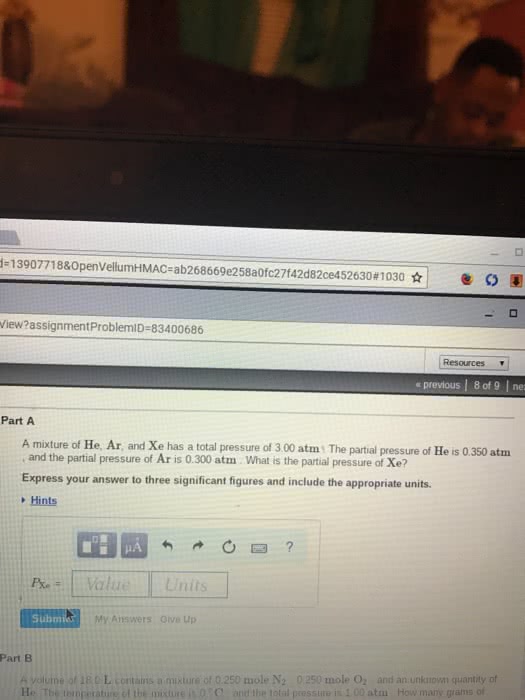
A volume of 18.8 L contains a mixture of 0.261 mole N2 , 0.257 mole O2 , and an unknown quantity of He. The temperature of the mixture is 0 °C , and the total pressure is 1.073 atm . How many grams of helium are present in the gas mixture?
A volume of 18.8 L contains a mixture of 0.261 mole N2 , 0.257 mole O2 , and an unknown quantity of He. The temperature of the mixture is 0 °C , and the total pressure is 1.073 atm . How many grams of helium are present in the gas mixture?