A solution is made by dissolving 15.4 g of cobalt(II) fluoride, CoF2, in enough water to make exactly 500 mL of solution. Calculate the concentration (molarity) of CoF2 in mol/L (M).
A solution is made by dissolving 15.4 g of cobalt(II) fluoride, CoF2, in enough water to make exactly 500 mL of solution. Calculate the concentration (molarity) of CoF2 in mol/L (M).
For unlimited access to Homework Help, a Homework+ subscription is required.
Related textbook solutions
Basic Chemistry
Principles of Chemistry Molecular Approach
Chemistry: Structure and Properties
Principles of Chemistry Molecular Approach
Chemistry: A Molecular Approach
Chemistry: A Molecular Approach
Principles of Chemistry: A Molecular Approach
Chemistry: The Central Science
Related questions
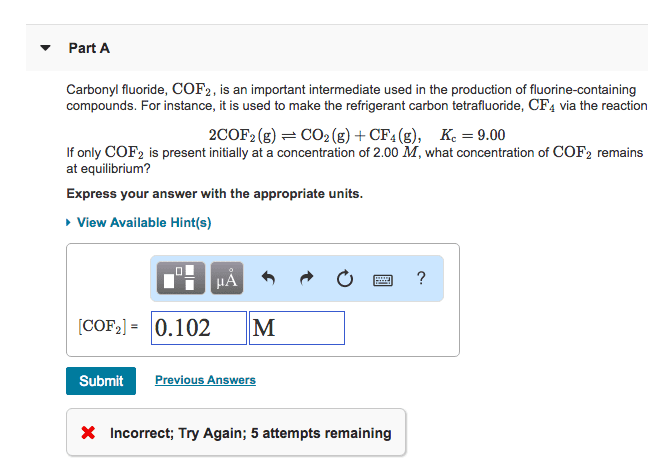
Part A
Carbonyl fluoride, COF2, is an important intermediate used in the production of fluorine-containing compounds. For instance, it is used to make the refrigerant carbon tetrafluoride, CF4 via the reaction
2COF2(g)âCO2(g)+CF4(g), Kc=5.90
If only COF2 is present initially at a concentration of 2.00 M, what concentration of COF2 remains at equilibrium?
| | |||
| [COF2] = |
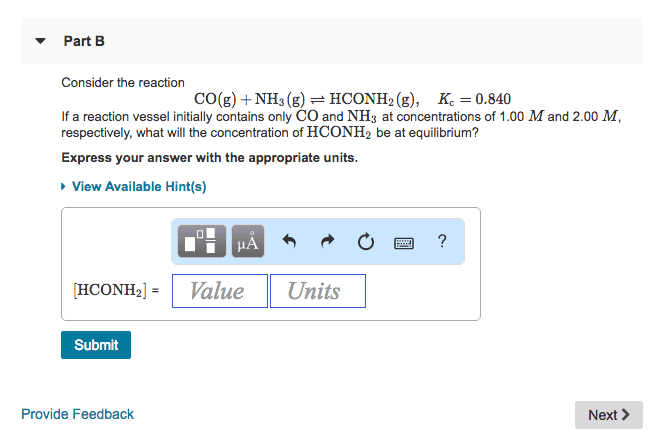
Part B
Consider the reaction
CO(g)+NH3(g)âHCONH2(g), Kc=0.750
If a reaction vessel initially contains only CO and NH3 at concentrations of 1.00 M and 2.00 M, respectively, what will the concentration of HCONH2 be at equilibrium?
| | |||
| [HCONH2] = |
Carbonyl fluoride, COF2, is an important intermediate used in the production of fluorine-containing compounds. For instance, it is used to make the refrigerant carbon tetrafluoride, CF4 via the reaction
2COF2(g)âCO2(g)+CF4(g), Kc=5.60
If only COF2 is present initially at a concentration of 2.00 M, what concentration of COF2 remains at equilibrium?
Express your answer with the appropriate unit
| | |||
| [COF2] = |
Part B
Consider the reaction
CO(g)+NH3(g)âHCONH2(g), Kc=0.740
If a reaction vessel initially contains only CO and NH3 at concentrations of 1.00 M and 2.00 M, respectively, what will the concentration of HCONH2 be at equilibrium?
Express your answer with the appropriate units.
| | |||
| [HCONH2] = |