1
answer
0
watching
2,783
views
13 Dec 2019
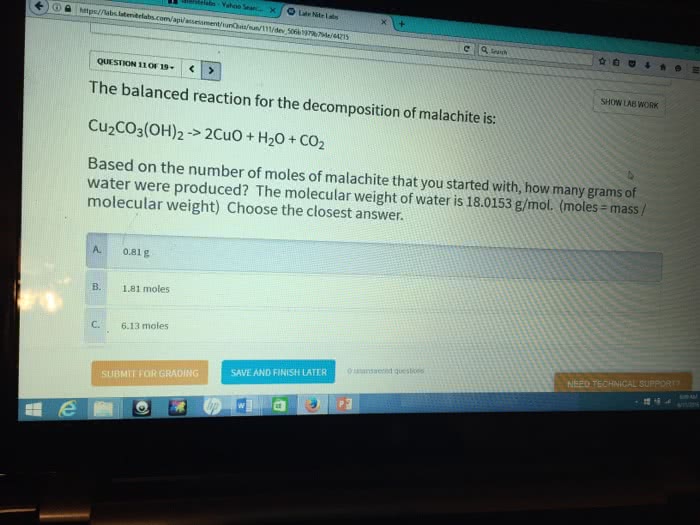
Based on the balanced chemical equation of Malachite: 2CuSO4(aq) +H2O + 2 Na2CO3(aq) -> CuCO3Cu(OH)2(s) + 2Na2SO4(aq)+CO2(g)
How many grams of malachite should be formed when 1.5 moles of copper sulfate reacts completly with an excess of sodium carbonate ?
Based on the balanced chemical equation of Malachite: 2CuSO4(aq) +H2O + 2 Na2CO3(aq) -> CuCO3Cu(OH)2(s) + 2Na2SO4(aq)+CO2(g)
How many grams of malachite should be formed when 1.5 moles of copper sulfate reacts completly with an excess of sodium carbonate ?
Jamar FerryLv2
17 Dec 2019