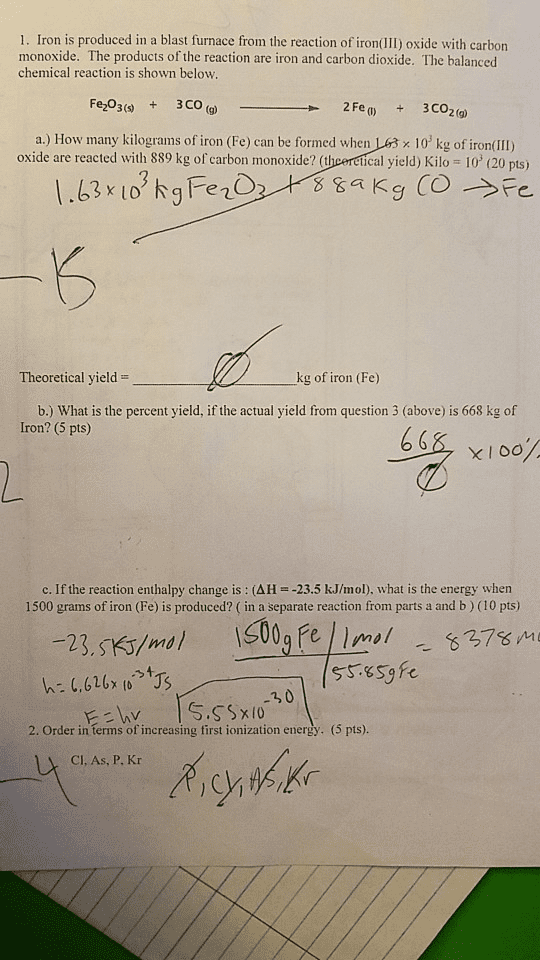1
answer
0
watching
1,201
views
13 Dec 2019
Iron(III) oxide, Fe2O3, otherwise known as rust, can react with pure carbon to reform solid iron and carbon monoxide. A balanced chemical equation for this reaction is included below. Fe2O3(s) + 3C(s) ? 2Fe(s) + 3CO(g)
a) Assuming you have 635 g of iron(III) oxide and 185 g of carbon, which of these reagents would be considered the limiting reagent? You must show all your work, including dimensional analysis, to receive credit for this problem!
b) What is the theoretical yield of Fe for this reaction (in g)?
Iron(III) oxide, Fe2O3, otherwise known as rust, can react with pure carbon to reform solid iron and carbon monoxide. A balanced chemical equation for this reaction is included below. Fe2O3(s) + 3C(s) ? 2Fe(s) + 3CO(g)
a) Assuming you have 635 g of iron(III) oxide and 185 g of carbon, which of these reagents would be considered the limiting reagent? You must show all your work, including dimensional analysis, to receive credit for this problem!
b) What is the theoretical yield of Fe for this reaction (in g)?
Casey DurganLv2
17 Dec 2019