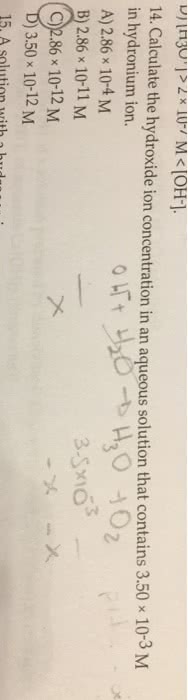1
answer
0
watching
168
views
13 Dec 2019
Calculate the H+ concentration in an aqueous solution that contains 2.50 x 10-3 M hydroxide ion.
a. 4.00 x 10-10
b. 4.00 x 10-11
c. 5.00 x 10-12
d. 4.00 x 10-12
Calculate the H+ concentration in an aqueous solution that contains 2.50 x 10-3 M hydroxide ion.
| a. | 4.00 x 10-10 | |
| b. | 4.00 x 10-11 | |
| c. | 5.00 x 10-12 | |
| d. | 4.00 x 10-12 |
Irving HeathcoteLv2
17 Dec 2019