1
answer
0
watching
189
views
13 Dec 2019
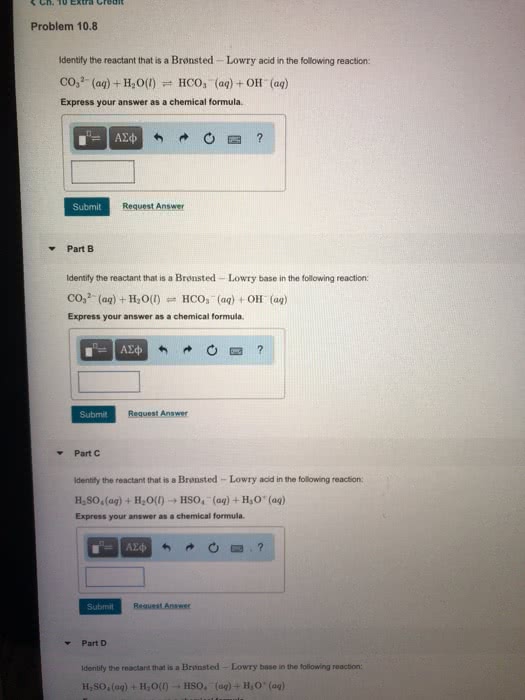
Part 1: For the following reaction, select all acid-base theories that apply. H2O(l) + :NH3(g) ? NH4+(aq) + OH-(aq)
A Arrhenius
B Brønsted-Lowry
C Lewis
Part 2: For the following reaction, select all acid-base theories that apply. :NH3(g) + BH3(g) ? NH3âBH3(s)
A Arrhenius
B Brønsted-Lowry
C Lewis
Part 3: For the following reaction, select all acid-base theories that apply. HCl(aq) + H2O(l) ? H3O+(aq) + Cl-(aq)
A Arrhenius
B Brønsted-Lowry
C Lewis
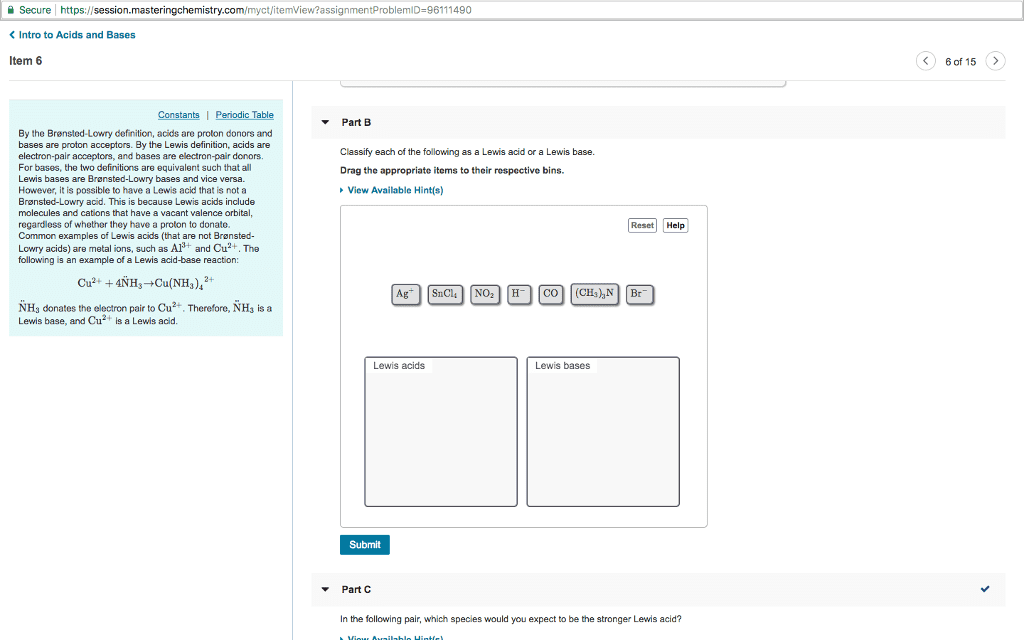
Part 4: Which of the following substances are amphoteric (can act as either a Brønsted-Lowry acid or base)?
A HCO3-
B H2PO4-
C NH4+
D H2O
E HS-
Part 1: For the following reaction, select all acid-base theories that apply. H2O(l) + :NH3(g) ? NH4+(aq) + OH-(aq)
A Arrhenius
B Brønsted-Lowry
C Lewis
Part 2: For the following reaction, select all acid-base theories that apply. :NH3(g) + BH3(g) ? NH3âBH3(s)
A Arrhenius
B Brønsted-Lowry
C Lewis
Part 3: For the following reaction, select all acid-base theories that apply. HCl(aq) + H2O(l) ? H3O+(aq) + Cl-(aq)
A Arrhenius
B Brønsted-Lowry
C Lewis
Part 4: Which of the following substances are amphoteric (can act as either a Brønsted-Lowry acid or base)?
A HCO3-
B H2PO4-
C NH4+
D H2O
E HS-
Patrina SchowalterLv2
17 Dec 2019