1
answer
0
watching
72
views
13 Dec 2019
Please help!
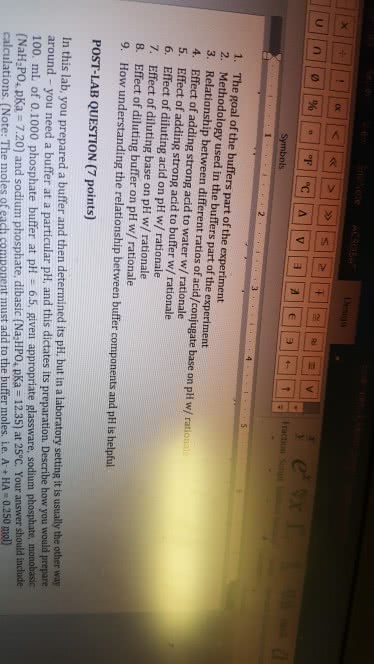
Describe how one would prepare in the laboratory 1.00 liter of a 50 millimolar sodium phosphate buffer solution having a ph of 7.0. The pKa values of phosphoric acid, H3PO4, are 2.14, 6.86, and 12.4. Assume reagent grade 80% (w/w) phosphoric acid solution and a standardized 0.100 M solution of NaOH available and needed glassware.
Please help!
Describe how one would prepare in the laboratory 1.00 liter of a 50 millimolar sodium phosphate buffer solution having a ph of 7.0. The pKa values of phosphoric acid, H3PO4, are 2.14, 6.86, and 12.4. Assume reagent grade 80% (w/w) phosphoric acid solution and a standardized 0.100 M solution of NaOH available and needed glassware.
1
answer
0
watching
72
views
For unlimited access to Homework Help, a Homework+ subscription is required.
Trinidad TremblayLv2
17 Dec 2019
Related textbook solutions
Basic Chemistry
5 Edition,
Timberlake
ISBN: 9780134138046
Principles of Chemistry Molecular Approach
4th Edition,
Tro
ISBN: 9780134112831
Chemistry: Structure and Properties
2nd Edition,
Tro
ISBN: 9780134293936
Principles of Chemistry Molecular Approach
3rd Edition, 2014
Tro
ISBN: 9780321971944
Chemistry: A Molecular Approach
3rd Edition,
Tro
ISBN: 9780321809247
Chemistry: A Molecular Approach
5th Edition,
Tro
ISBN: 9780134874371
Principles of Chemistry: A Molecular Approach
4th Edition,
Tro
ISBN: 9780134895741
Chemistry: The Central Science
14th Edition, 2017
Brown
ISBN: 9780134414232