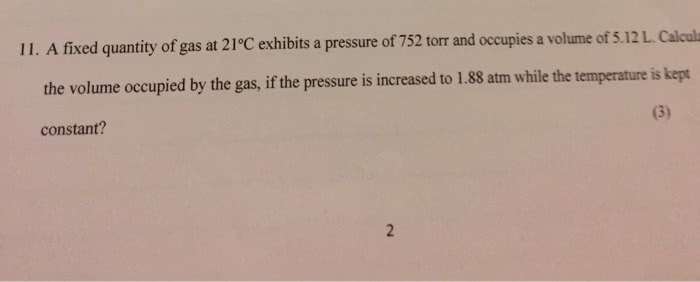1
answer
0
watching
204
views
13 Dec 2019
A fixed quantity of gas at 21 âC exhibits a pressure of 755 torr and occupies a volume of 5.02 L . Calculate the volume the gas will occupy if the pressure is increased to 1.85 atm while the temperature is held constant.
A fixed quantity of gas at 21 âC exhibits a pressure of 755 torr and occupies a volume of 5.02 L . Calculate the volume the gas will occupy if the pressure is increased to 1.85 atm while the temperature is held constant.
Jarrod RobelLv2
17 Dec 2019