1
answer
0
watching
149
views
13 Dec 2019
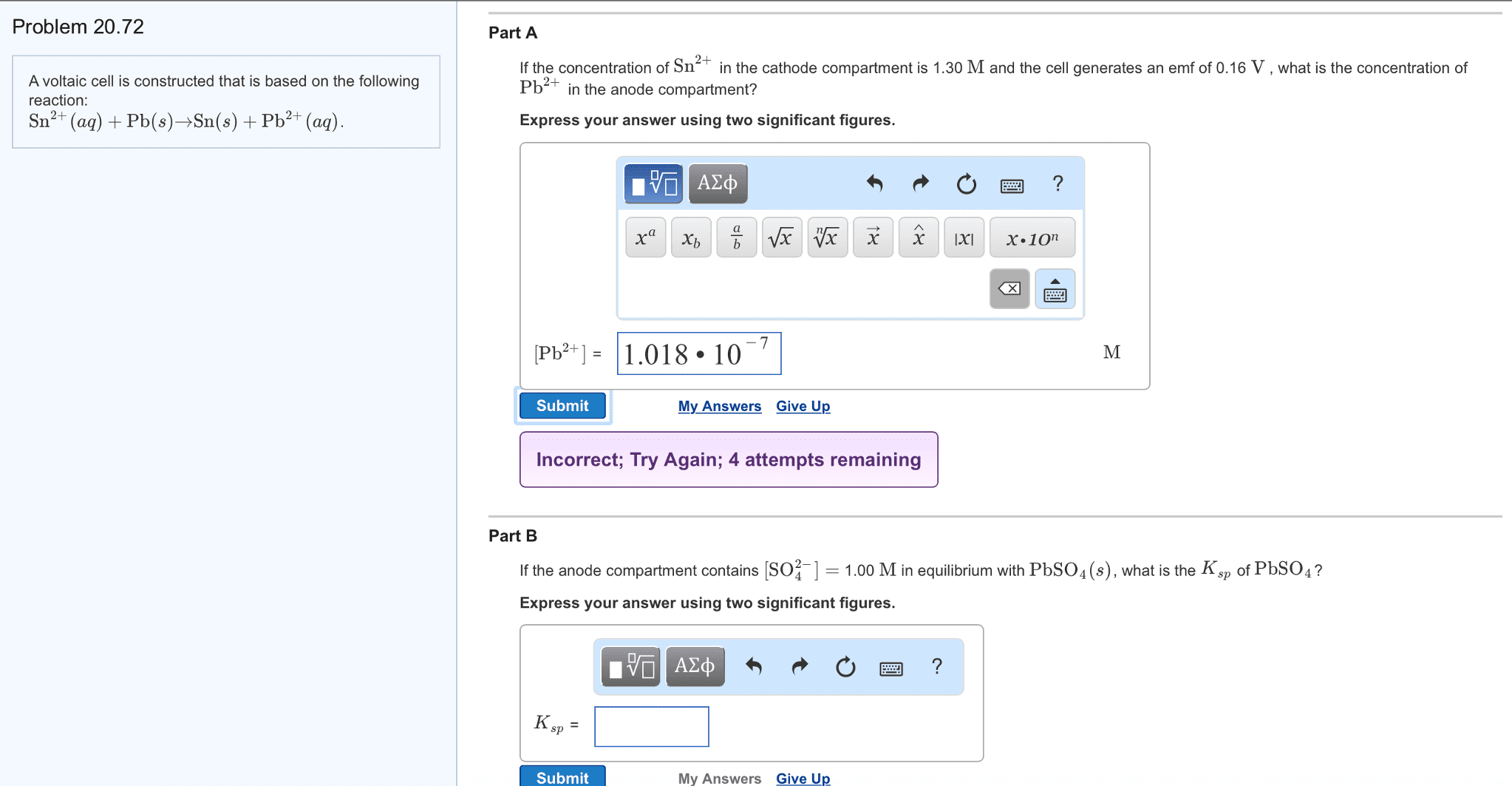
A concentration cell similar to the one shown is composed of two Fe electrodes and solutions of different Fe2+concentrations. The left compartment contains 0.477 M Fe2+, and the right compartment contains 0.450 M Fe2+.
Calculate the cell potential for this reaction at 298 K.
____ volts
In this iron concentration cell, the compartment on the left is the _________ (anode or cathode) , and the compartment on the right is the _________ ( anode or cathode).
A concentration cell similar to the one shown is composed of two Fe electrodes and solutions of different Fe2+concentrations. The left compartment contains 0.477 M Fe2+, and the right compartment contains 0.450 M Fe2+.
Calculate the cell potential for this reaction at 298 K.
____ volts
In this iron concentration cell, the compartment on the left is the _________ (anode or cathode) , and the compartment on the right is the _________ ( anode or cathode).
Lelia LubowitzLv2
17 Dec 2019