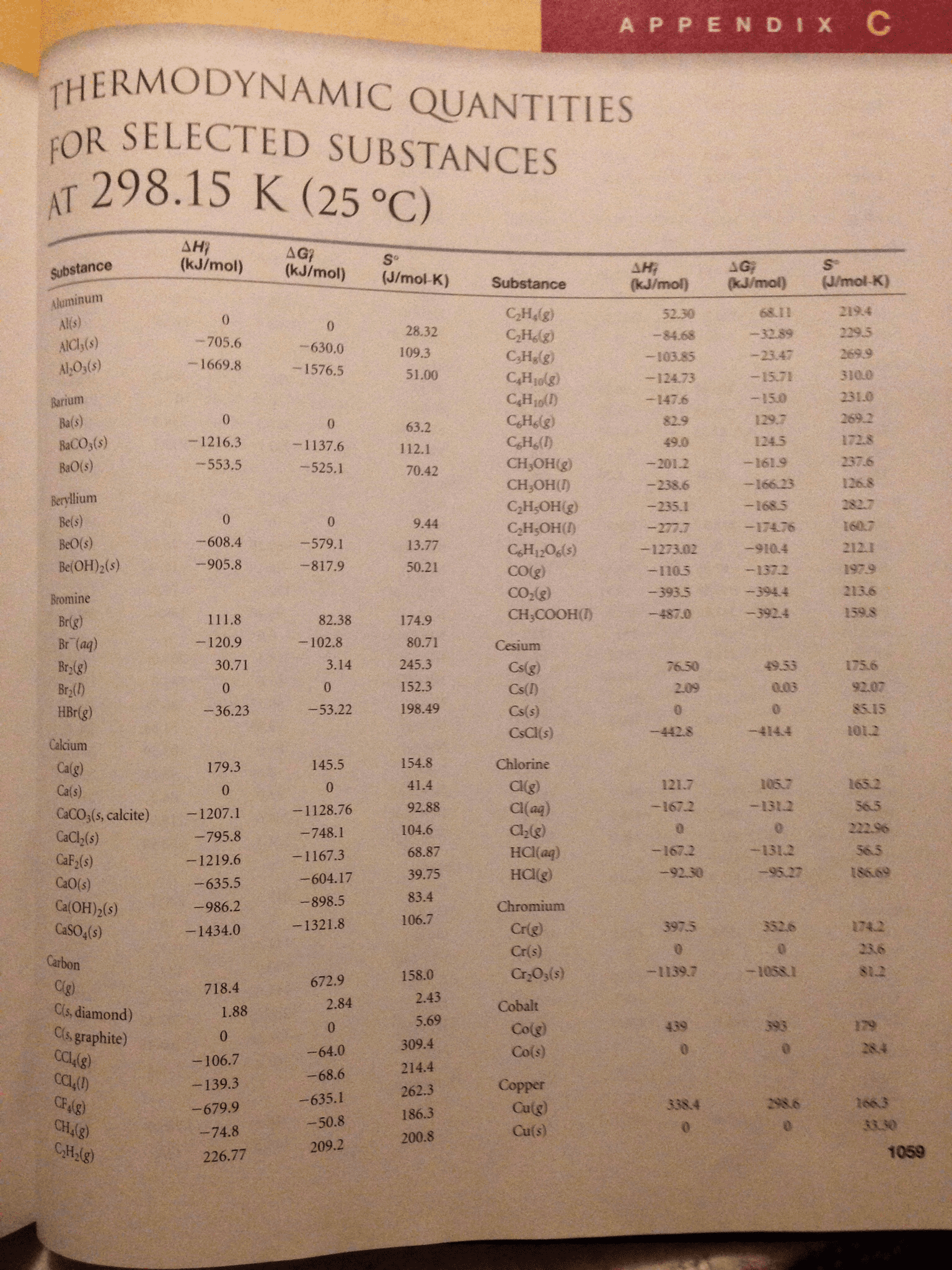
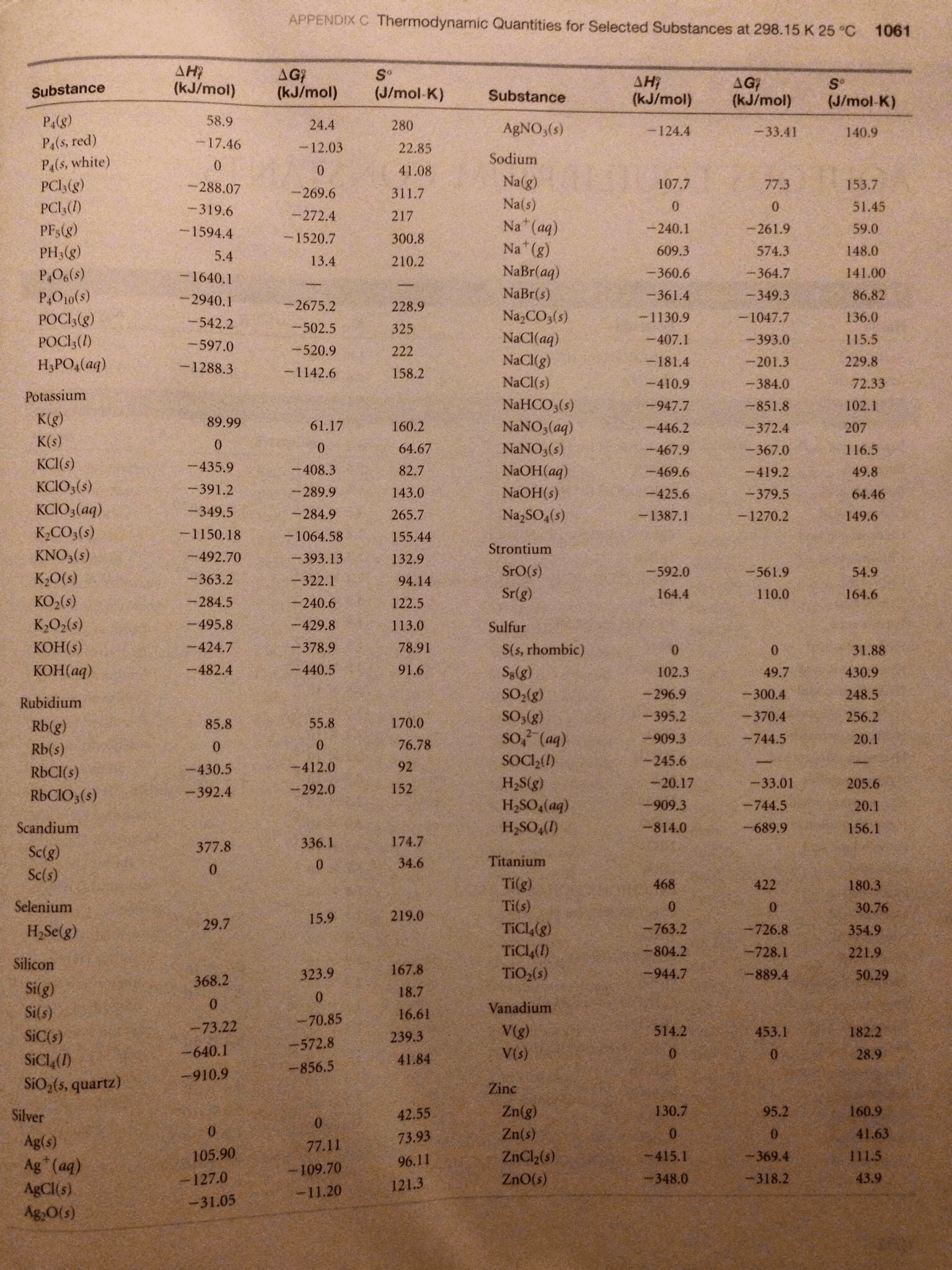
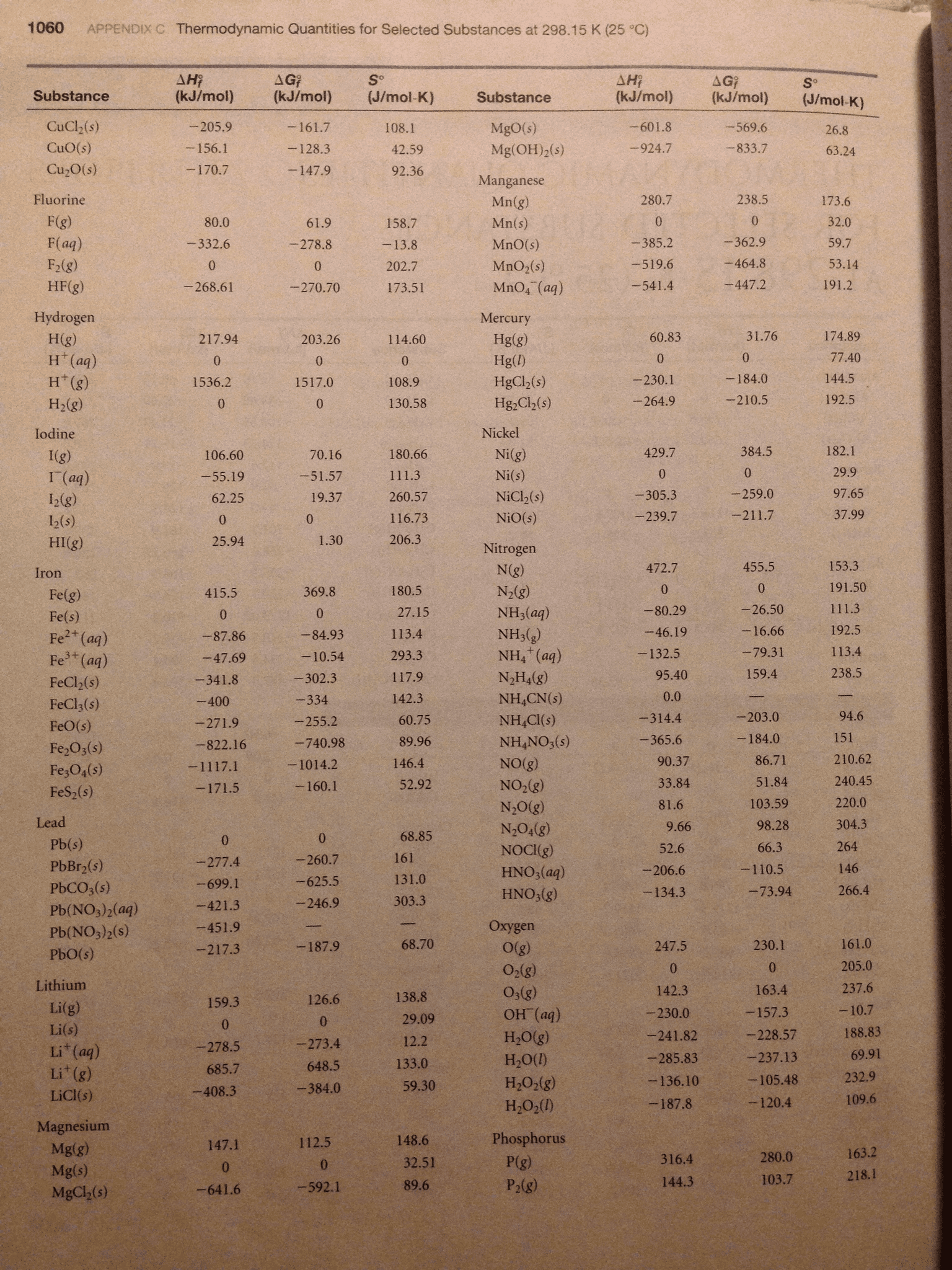
1. You will investigate three reactions in this experiment. In the space provided below, write the balanced molecular and net ionic equations from the descriptions of the reactions. Use the table of standard heats of formation in your text (or another approved resource) to calculate the molar enthalpies of the reactions.
Reaction 1: An aqueous solution of sodium hydroxide reacts with an aqueous solution of hydrochloric acid, yielding water.
Reaction 2: An aqueous solution of sodium hydroxide reacts with an aqueous solution of ammonium chloride, yielding aqueous ammonia, NH3, and water.
Reaction 3: An aqueous solution of hydrochloric acid reacts with aqueous ammonia, NH3, yielding aqueous ammonium chloride.
Reaction
Balanced reaction equation
ÎH (kJ)
1
2
3
2. For each reaction you perform, you will mix together 50.0 mL of 2.00 M acid and 2.00 M base. What are the resulting concentrations of the reactants upon mixing?
3. How many moles of each reactant are present in each trial?
4. What is the total volume of the solutions upon mixing? Assuming a density of 1.03 g/mL for the solution, find the total mass of each reaction mixture.
1. You will investigate three reactions in this experiment. In the space provided below, write the balanced molecular and net ionic equations from the descriptions of the reactions. Use the table of standard heats of formation in your text (or another approved resource) to calculate the molar enthalpies of the reactions.
Reaction 1: An aqueous solution of sodium hydroxide reacts with an aqueous solution of hydrochloric acid, yielding water.
Reaction 2: An aqueous solution of sodium hydroxide reacts with an aqueous solution of ammonium chloride, yielding aqueous ammonia, NH3, and water.
Reaction 3: An aqueous solution of hydrochloric acid reacts with aqueous ammonia, NH3, yielding aqueous ammonium chloride.
| Reaction | Balanced reaction equation | ÎH (kJ) |
| 1 | ||
| 2 | ||
| 3 |
2. For each reaction you perform, you will mix together 50.0 mL of 2.00 M acid and 2.00 M base. What are the resulting concentrations of the reactants upon mixing?
3. How many moles of each reactant are present in each trial?
4. What is the total volume of the solutions upon mixing? Assuming a density of 1.03 g/mL for the solution, find the total mass of each reaction mixture.