0
answers
0
watching
139
views
13 Dec 2019
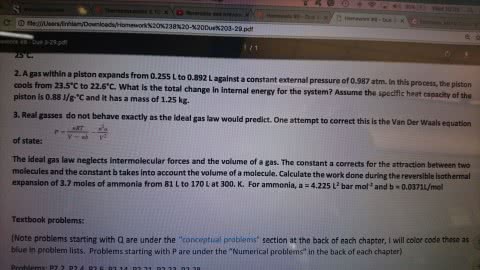
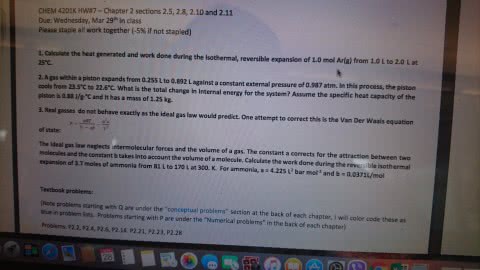
One mole of an ideal monatomic gas is inititally at 300 K and 1010 J/L pressure inside a cylinder with a frictionless piston. the cylinder is an isolated (adiabatic) system. the gas expands against zero external pressure. when the volume expands to 24.7 L, a peg stopes the piston. calculate deltaU (the change in energy), q (the heat transferred to the system), and w (the work done by the system) when the expansion is isothermal and reversible.
One mole of an ideal monatomic gas is inititally at 300 K and 1010 J/L pressure inside a cylinder with a frictionless piston. the cylinder is an isolated (adiabatic) system. the gas expands against zero external pressure. when the volume expands to 24.7 L, a peg stopes the piston. calculate deltaU (the change in energy), q (the heat transferred to the system), and w (the work done by the system) when the expansion is isothermal and reversible.