1
answer
0
watching
127
views
13 Dec 2019
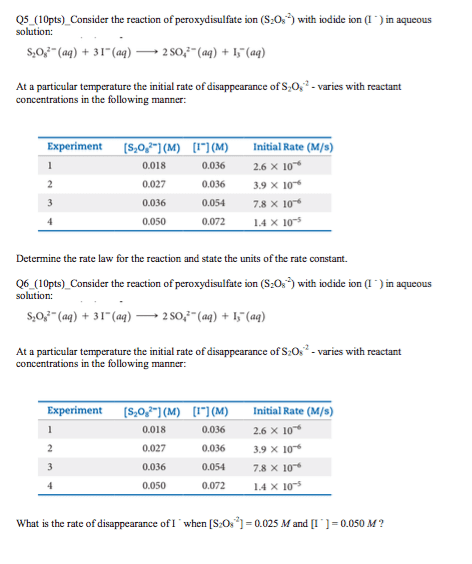
Consider the reaction of peroxydisulfate ion (S2O2?8) with iodide ion (I?) in aqueous solution:
S2O2?8(aq)+3I?(aq)?2SO2?4(aq)+I?3(aq).
At a particular temperature the rate of disappearance of S2O2?8 varies with reactant concentrations in the following manner:
Experiment S2O2?8(M) I?(M) Initial Rate (M/s) 1 0.018 0.036 2.6Ã10?6 2 0.027 0.036 3.9Ã10?6 3 0.036 0.054 7.8Ã10?6 4 0.050 0.072 1.4Ã10?5
Part D - What is the rate of disappearance of I? when [S2O2?8]=2.8Ã10?2 M and [I?]= 5.0Ã10?2 M ?
Consider the reaction of peroxydisulfate ion (S2O2?8) with iodide ion (I?) in aqueous solution:
S2O2?8(aq)+3I?(aq)?2SO2?4(aq)+I?3(aq).
At a particular temperature the rate of disappearance of S2O2?8 varies with reactant concentrations in the following manner:
| Experiment | S2O2?8(M) | I?(M) | Initial Rate (M/s) |
| 1 | 0.018 | 0.036 | 2.6Ã10?6 |
| 2 | 0.027 | 0.036 | 3.9Ã10?6 |
| 3 | 0.036 | 0.054 | 7.8Ã10?6 |
| 4 | 0.050 | 0.072 | 1.4Ã10?5 |
Part D - What is the rate of disappearance of I? when [S2O2?8]=2.8Ã10?2 M and [I?]= 5.0Ã10?2 M ?
Nestor RutherfordLv2
17 Dec 2019