1
answer
0
watching
396
views
13 Dec 2019
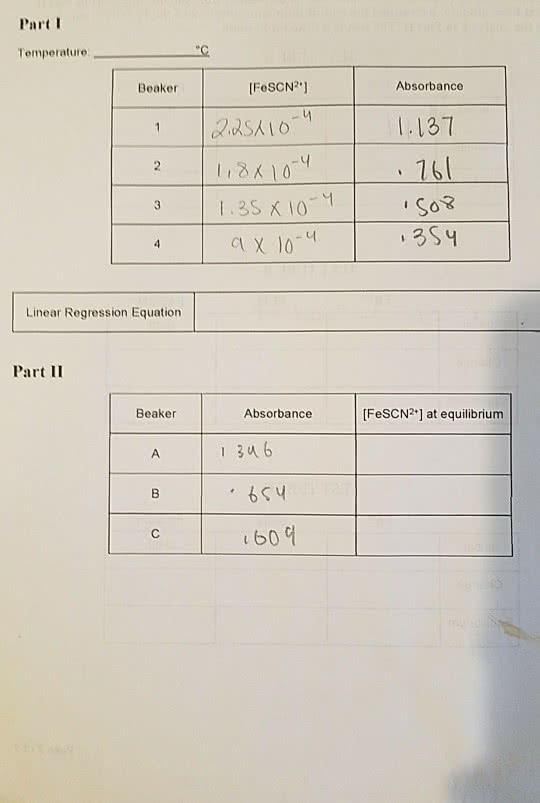
Consider the following reaction: Fe3+ (aq) + SCN- (aq) <--> FeSCN2+ (aq)
A 5.0 mL volume of 0.0200 M Fe(NO3)3 is mixed with 5.0 mL of 0.00200 M NaSCN; the blood-red [FeSCN2+] ion forms and an equilibrium is established between reactants and products. The molar concentration of [FeSCN2+] measured spectrophotometrically is determined to be 7.0 x 10-4 M at equilibrium. Calculate Kc showing all work and employing an ICE table.
Consider the following reaction: Fe3+ (aq) + SCN- (aq) <--> FeSCN2+ (aq)
A 5.0 mL volume of 0.0200 M Fe(NO3)3 is mixed with 5.0 mL of 0.00200 M NaSCN; the blood-red [FeSCN2+] ion forms and an equilibrium is established between reactants and products. The molar concentration of [FeSCN2+] measured spectrophotometrically is determined to be 7.0 x 10-4 M at equilibrium. Calculate Kc showing all work and employing an ICE table.
Bunny GreenfelderLv2
17 Dec 2019