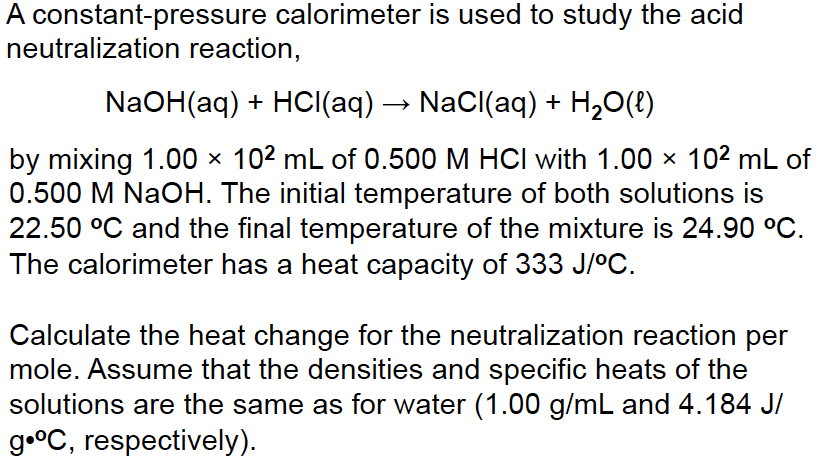1
answer
0
watching
93
views
13 Dec 2019
Consider the balanced neutralization reaction below, and answer the following questions:
HCl(aq) + NaOH(aq) ââââ> NaCl(aq) + H2O(l)
50.0 mL of 1.00 M HCl and 50.0 mL of 1.00 M NaOH, both at 22.5°C are added to a calorimeter and allowed to react and thermally equilibrate, raising the temperature of the calorimeter to 29.4°C. Assume that the total mass of water is 100.0 g. The specific heat of water = 4.184 J/g °C
How much total energy is transferred from the reactants to the water?
What is the molar heat of neutralization for the reaction in kJ/mol?
Consider the balanced neutralization reaction below, and answer the following questions:
HCl(aq) + NaOH(aq) ââââ> NaCl(aq) + H2O(l)
50.0 mL of 1.00 M HCl and 50.0 mL of 1.00 M NaOH, both at 22.5°C are added to a calorimeter and allowed to react and thermally equilibrate, raising the temperature of the calorimeter to 29.4°C. Assume that the total mass of water is 100.0 g. The specific heat of water = 4.184 J/g °C
How much total energy is transferred from the reactants to the water?
What is the molar heat of neutralization for the reaction in kJ/mol?
Beverley SmithLv2
17 Dec 2019