1
answer
0
watching
754
views
13 Dec 2019
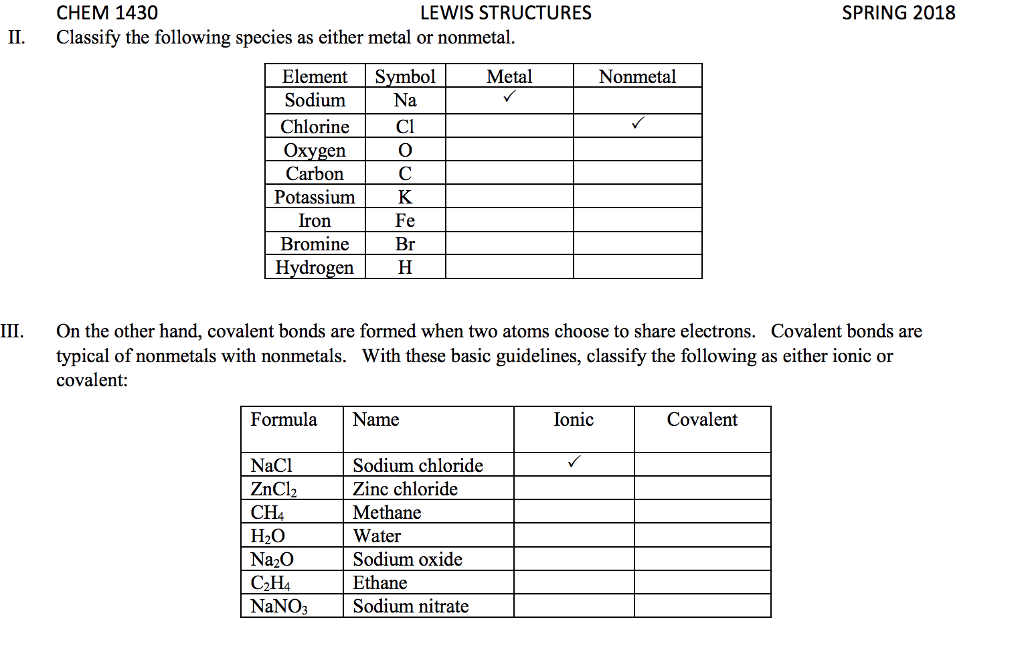
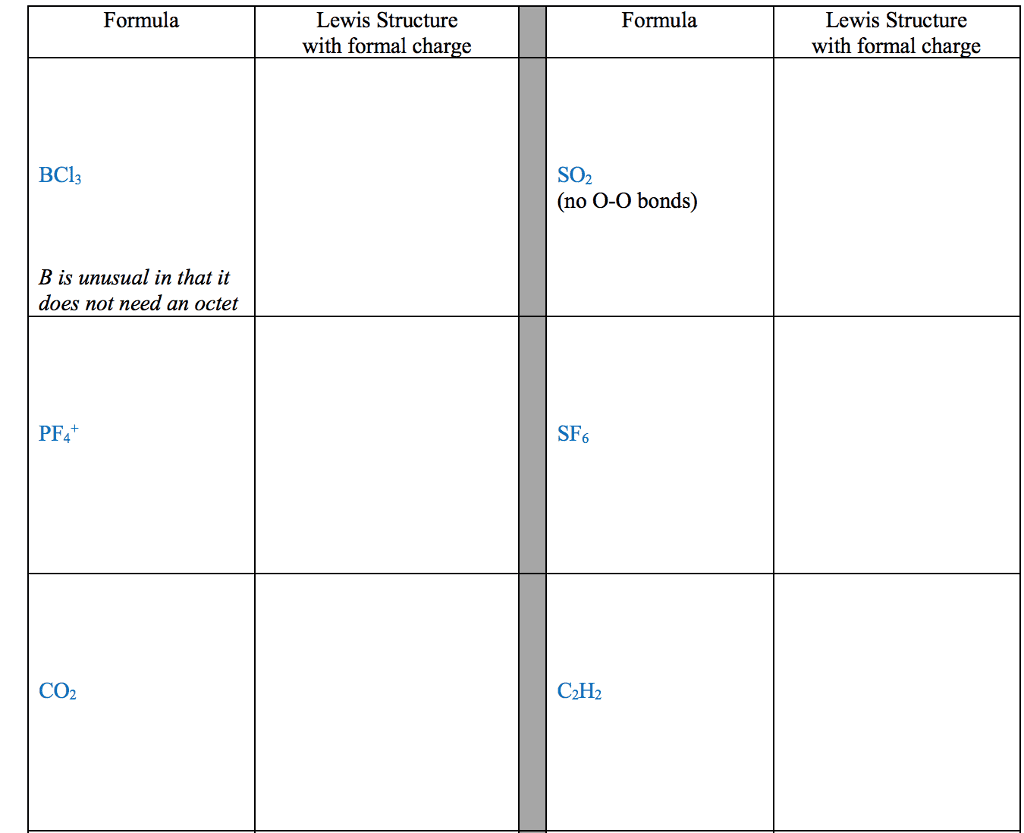
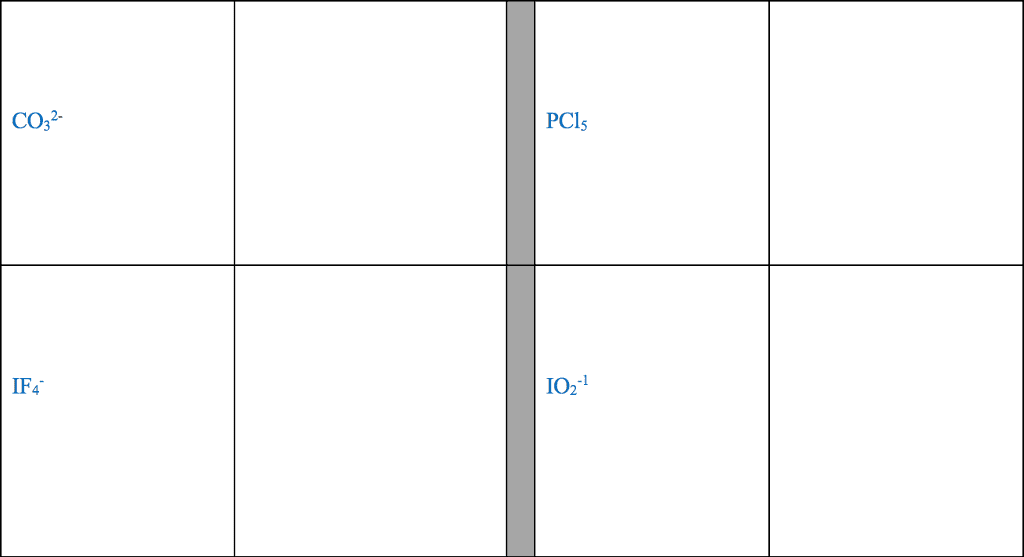
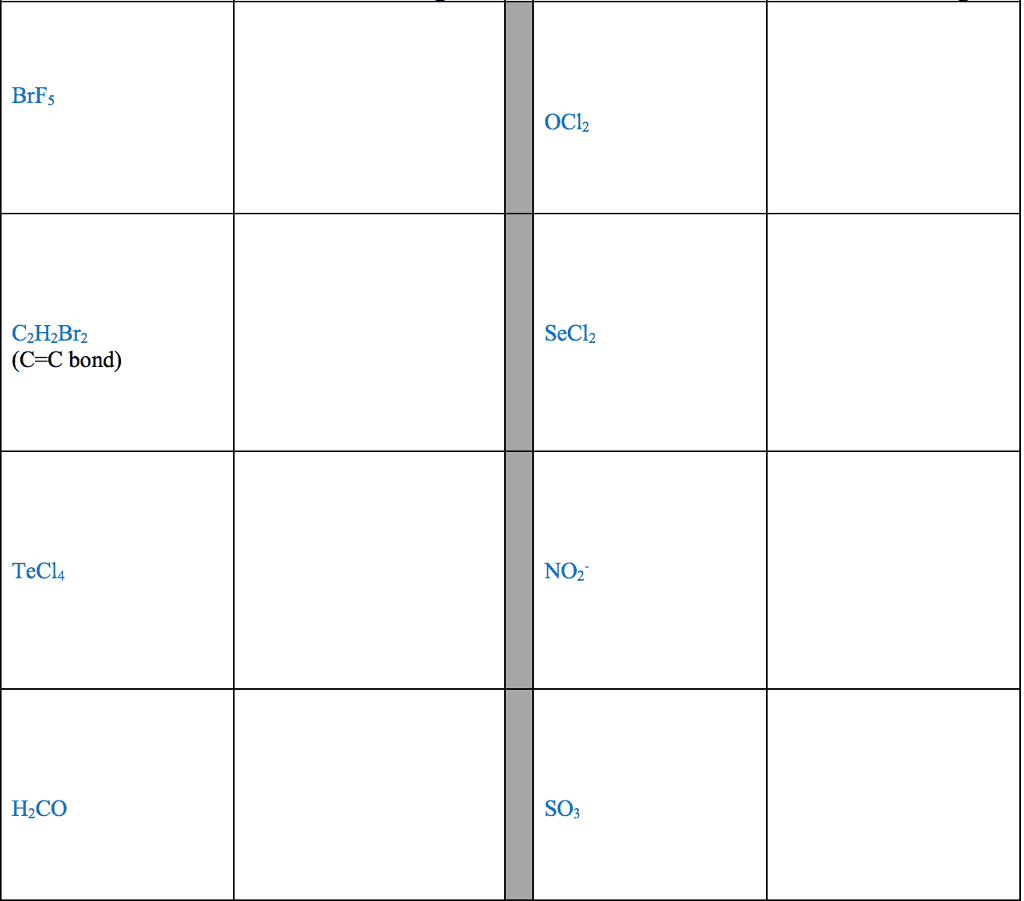
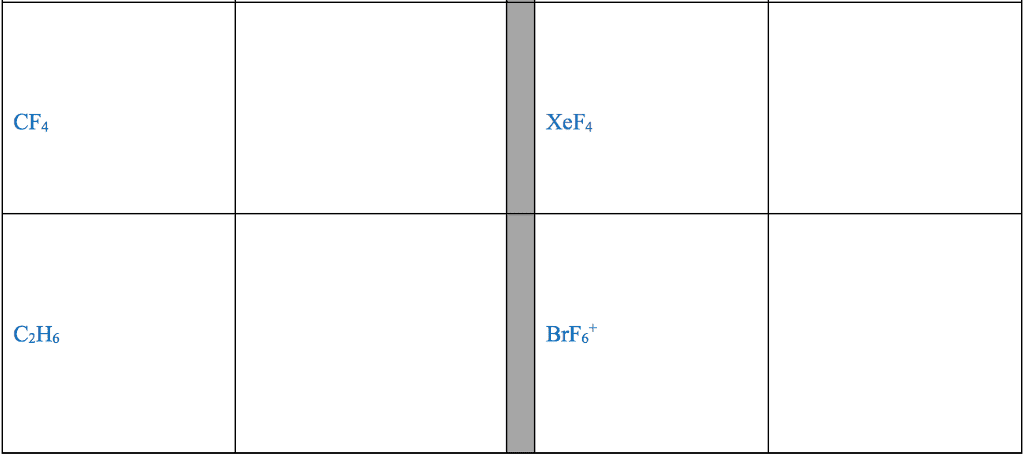
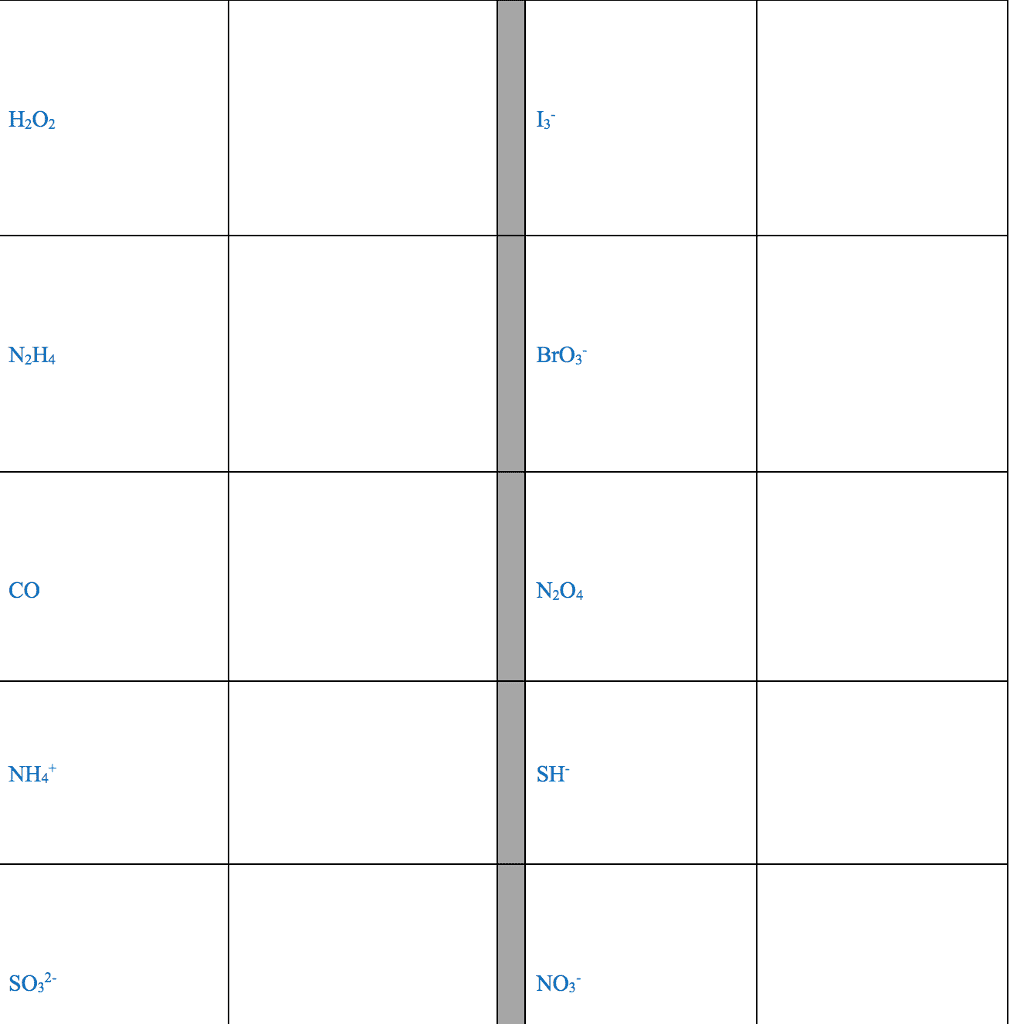
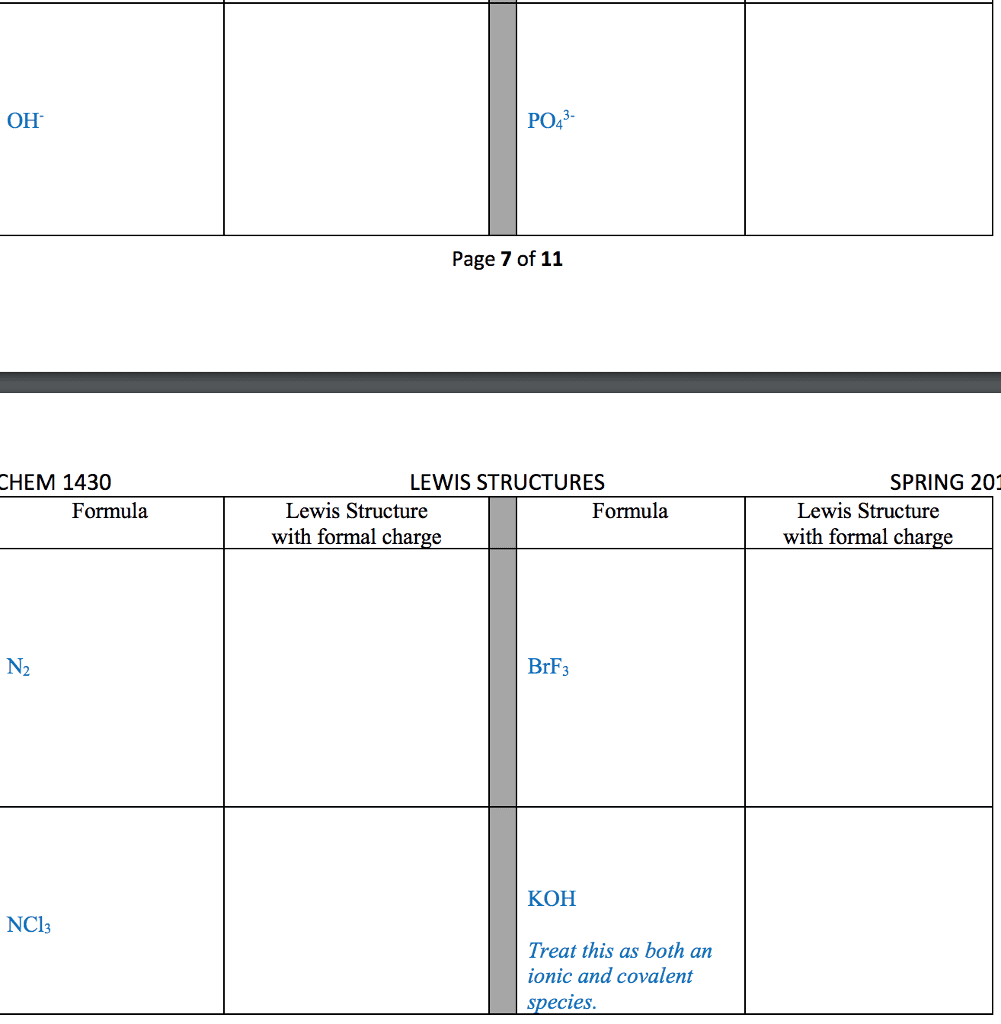
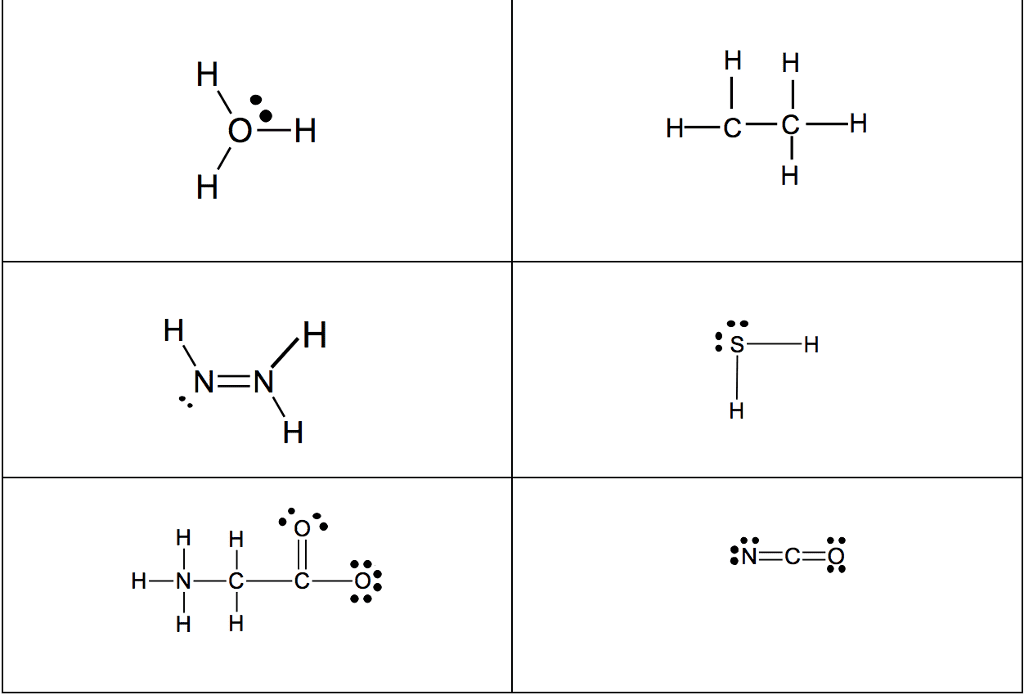
For each of the general electron-dot formula for elements, give thefollowing information [1] the number of valence electrons, [2] thegroup number of elements, [3] How many electrons would be gained orlost to achieved a noble gas configuration. [4] the charge on theresulting ons, [5] an example of the elements
1]. .Q. ( 2 dots)
2]. D ( has 5 dots)
3]. X (has 1 dot)
4]. A (has 7 dots)
.Q. : # of valence :Group # :# of electron : Charge : Example
: Electron :Ans: selection A & B) gain/lost : :
: : : : :
For each of the general electron-dot formula for elements, give thefollowing information [1] the number of valence electrons, [2] thegroup number of elements, [3] How many electrons would be gained orlost to achieved a noble gas configuration. [4] the charge on theresulting ons, [5] an example of the elements
1]. .Q. ( 2 dots)
2]. D ( has 5 dots)
3]. X (has 1 dot)
4]. A (has 7 dots)
.Q. : # of valence :Group # :# of electron : Charge : Example
: Electron :Ans: selection A & B) gain/lost : :
: : : : :
Liked by arsiachhetri14
Irving HeathcoteLv2
17 Dec 2019