1
answer
0
watching
149
views
13 Dec 2019
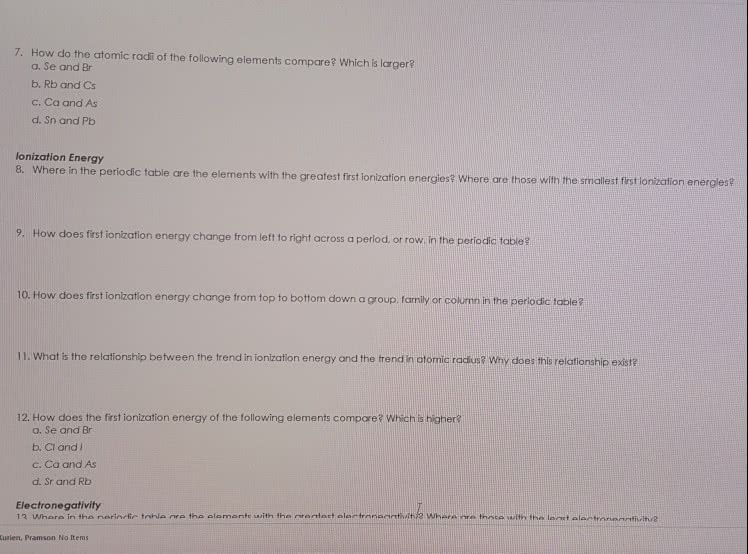
Arrange the members of the following set of elements in order of increasing first ionization energies. (Use the drop down lists to indicate your choice, with 1 as the smallest IE and 6 as the largest IE.)
Se
Te
In
O
Rb
S
Arrange the members of the following set of elements in order of increasing first ionization energies. (Use the drop down lists to indicate your choice, with 1 as the smallest IE and 6 as the largest IE.)
Se
Te
In
O
Rb
S
Casey DurganLv2
17 Dec 2019