COULD SOMEONE PLEASE HELP ME ANSWER THESE QUESTIONS AND IF POSSIBLE EXPLAIN?
1. Standard enthalpies of formation are usually computed from calorimetric data referring to reactions which are easy to carry out in the laboratory. For many compounds, the enthalpy of the reaction with O2 (g) (combustion) is commonly used, together with the standard enthalpies of formation of the combustions products, to compute ?Hf0. For example, sucrose, C12H22O11, common table sugar, is found to have a standard enthalpy of combustion of -5640.9 kJ mole-1. The standard enthalpies of formation of CO2 (g) and H2O (l) are, respectively, -393.51 and -285.83 kJ mole-1. Compute ?Hf0 for sucrose.
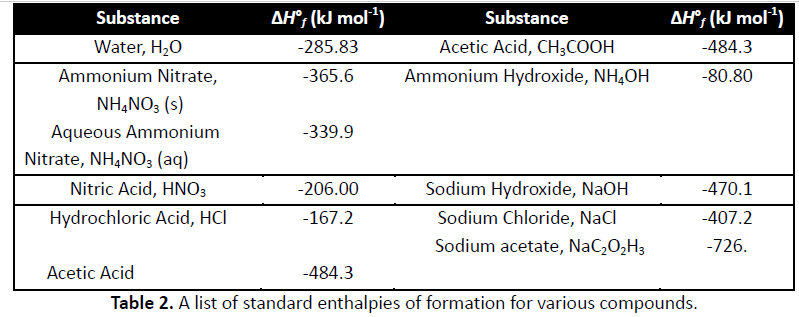
2. A student determined the enthalpy of solution of NaOH by dissolving 4.00 g of NaOH (s) in 180 ml of pure water (density 0.999 g ml-1) at an initial temperature of 19.5
COULD SOMEONE PLEASE HELP ME ANSWER THESE QUESTIONS AND IF POSSIBLE EXPLAIN?
1. Standard enthalpies of formation are usually computed from calorimetric data referring to reactions which are easy to carry out in the laboratory. For many compounds, the enthalpy of the reaction with O2 (g) (combustion) is commonly used, together with the standard enthalpies of formation of the combustions products, to compute ?Hf0. For example, sucrose, C12H22O11, common table sugar, is found to have a standard enthalpy of combustion of -5640.9 kJ mole-1. The standard enthalpies of formation of CO2 (g) and H2O (l) are, respectively, -393.51 and -285.83 kJ mole-1. Compute ?Hf0 for sucrose.
2. A student determined the enthalpy of solution of NaOH by dissolving 4.00 g of NaOH (s) in 180 ml of pure water (density 0.999 g ml-1) at an initial temperature of 19.5