1
answer
0
watching
192
views
13 Dec 2019
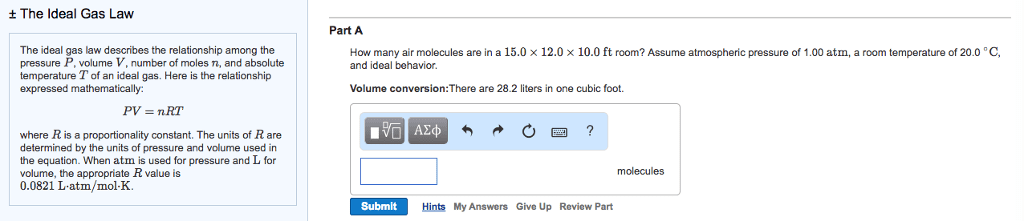
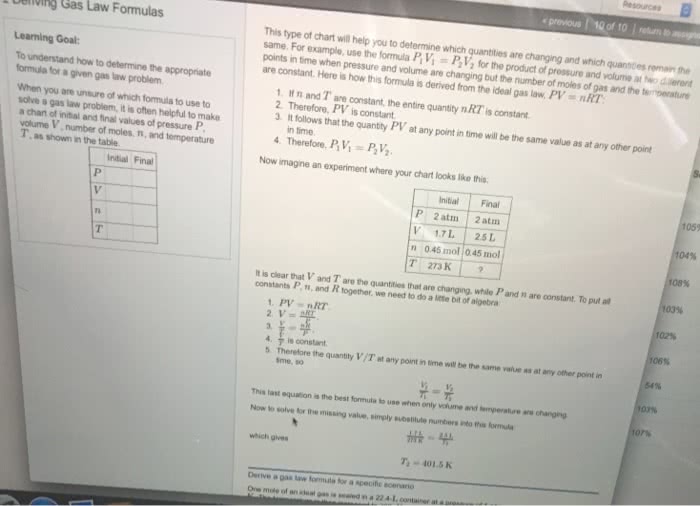
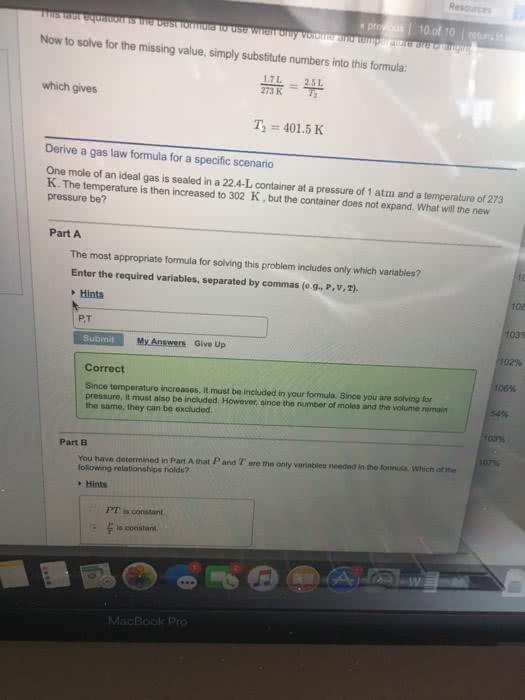
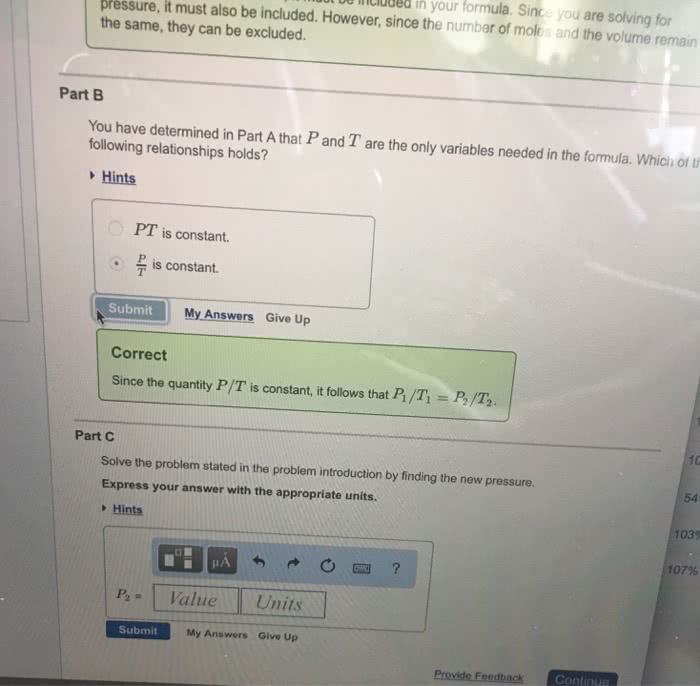
Suppose that the equation of state of a real gas is given by
PV = nRT + na(P/T)
where a = 4.0 L K/mol is a constant. Compute the entropy change upon changing the pressure of 1 mole of gas from 2 atm to 1 atm at a const. temperature of 300K. Compare this to the entropy change for an ideal gas under the same conditions.
Suppose that the equation of state of a real gas is given by
PV = nRT + na(P/T)
where a = 4.0 L K/mol is a constant. Compute the entropy change upon changing the pressure of 1 mole of gas from 2 atm to 1 atm at a const. temperature of 300K. Compare this to the entropy change for an ideal gas under the same conditions.
1
answer
0
watching
192
views
For unlimited access to Homework Help, a Homework+ subscription is required.
Jamar FerryLv2
17 Dec 2019
Related textbook solutions
Basic Chemistry
5 Edition,
Timberlake
ISBN: 9780134138046
Principles of Chemistry Molecular Approach
4th Edition,
Tro
ISBN: 9780134112831
Chemistry: Structure and Properties
2nd Edition,
Tro
ISBN: 9780134293936
Principles of Chemistry Molecular Approach
3rd Edition, 2014
Tro
ISBN: 9780321971944
Chemistry: A Molecular Approach
3rd Edition,
Tro
ISBN: 9780321809247
Chemistry: A Molecular Approach
5th Edition,
Tro
ISBN: 9780134874371
Principles of Chemistry: A Molecular Approach
4th Edition,
Tro
ISBN: 9780134895741
Chemistry: The Central Science
14th Edition, 2017
Brown
ISBN: 9780134414232