0
answers
0
watching
198
views
13 Dec 2019
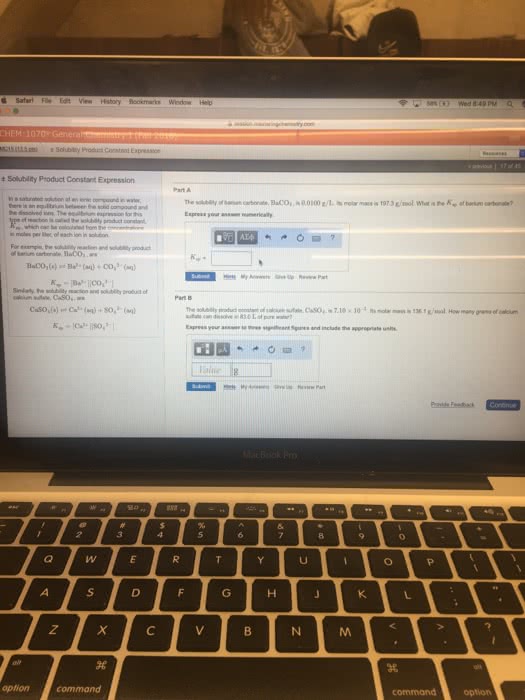
1) Pb3(AsO4)2 is slightly soluble in water, if the solubility is 3,0*10^-5 g/L, what is the solubility product constant? Assume that the solubility equilibrium is the only important one.
2) Anhydrite is a calcium sulfate mineral deposited when seawater evaporates. what is the solubility of calcium sulfate in grams per liter? The solubility product constant for calcium sulfate is 2.4*10^-5.
3) Calculate the molar solubility of barium fluoride in water at 25 degree C. the solubility product constant is 1.0*10^-6.
1) Pb3(AsO4)2 is slightly soluble in water, if the solubility is 3,0*10^-5 g/L, what is the solubility product constant? Assume that the solubility equilibrium is the only important one.
2) Anhydrite is a calcium sulfate mineral deposited when seawater evaporates. what is the solubility of calcium sulfate in grams per liter? The solubility product constant for calcium sulfate is 2.4*10^-5.
3) Calculate the molar solubility of barium fluoride in water at 25 degree C. the solubility product constant is 1.0*10^-6.